Abstract.
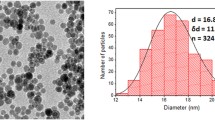
The purpose of this study was to apply a new cell culture assay that preserves hepatocyte orientation and differentiation for screening of MR contrast agents with hepatocyte specificity. Cultured hepatocytes were sandwiched between two layers of collagen, preserving both hepatocyte function and morphology over a prolonged period of time. Plain and rhodaminated monocrystalline iron-oxide particles (MION and MION-rh) and asialoglycoprotein receptor-specific rhodaminated asialofetuin coupled to MION (MION-ASF-rh) were prepared. Dose-dependent competition experiments of these agents were performed with D( + )-galactose to determine the specificity of galactose-mediated cell uptake. To assess the impact of cell integrity on cell uptake dose-dependent functional experiments with two hepatotoxins (ethanol and CCl4) were performed. Normal cell cultures showed significantly higher fluorescent-light emission after incubation with hepatocyte-directed ASF-MION-rh than after incubation with MION-rh. Competition experiments of ASF-MION with galactose showed a dose-dependent decrease in calibrated fluorescent-light emission. Cell cultures treated with hepatotoxins demonstrated a dose-dependent reduction in calibrated fluorescent-light emission following incubation with ASF-MION-rh. The validated assay system allows assessment not only of hepatocyte specificity, but also of hepatocyte damage. Because the assay can be applied to cells from any species (rat, pig, human), it may represent an ideal test system prior to clinical trials of new hepatocyte-directed MR contrast agents.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received 31 July 1996; Revision received 25 September 1996; Accepted 26 September 1996
Rights and permissions
About this article
Cite this article
Reimer, P., Bader, A. & Weissleder, R. Application of a stable cell culture assay for the functional assessment of novel MR contrast agents. Eur Radiol 7, 527–531 (1997). https://doi.org/10.1007/s003300050197
Issue Date:
DOI: https://doi.org/10.1007/s003300050197