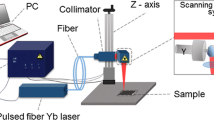
2
O3. Based on Auger Electron Spectroscopy analyses and optical microscopic observations, γ-Fe2O3 and Fe3O4 are formed on the stainless steel surface in laser cleaning in the air. Since laser can induce high temperature rise in stainless steel surfaces, the above phenomenon can be explained by a thermochemical reaction between oxygen in the air and the stainless steel. With increasing laser fluences, the temperature rise in the irradiated area of stainless steel surface increases, which enhances oxygen diffusion into the surface and oxidation reaction within the irradiated area. In order to avoid discoloration of stainless steel surfaces, a vacuum system was used to reduce the oxidation reaction between oxygen and stainless steel.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 7 June 1996/Accepted: 30 September 1996
Rights and permissions
About this article
Cite this article
Lu, Y., Song, W., Hong, M. et al. Mechanism of and method to avoid discoloration of stainless steel surfaces in laser cleaning . Appl Phys A 64, 573–578 (1997). https://doi.org/10.1007/s003390050518
Issue Date:
DOI: https://doi.org/10.1007/s003390050518