Summary
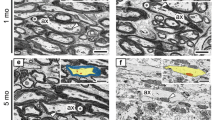
Morphological alterations of oligodendroglia were investigated in the spinal cord of the twitcher mouse, an authentic murine model of human globoid cell leukodystrophy (GLD) from day 5 to day 45 postnatal (p.n.).
Typical inclusions were seen in the perikarya as well as the processes of oligodendroglia after day 10 with increasing frequency. The majority of the inclusions was non-crystalloid but rather needle-like or slender tubular in appearance. Ultrastructural features of cellular degeneration became first noticeable on days 25–30 in the oligodendroglial cytoplasm. These consisted of an increased number of microtubules and/or smooth cisterns, dispersed ribosomes, alteration of endoplasmic reticulum forming stacked lamellae or whorles, vesiculation or vacuolation of cytoplasm. The number of degenerating oligodendroglia increased in the older twitcher mice, so did the degenerating myelin sheath. However, even on day 45, when globoid cells became conspicuous in subpial and perivascular regions, many oligodendroglia and myelin sheaths were still well preserved.
These observations suggested that oligodendrogial degeneration resulted in the degeneration of myelin sheaths but globoid cells appeared even before morphological evidence of myelin degeneration, presumably in response to the biochemical alterations resulted from the deficiency of galactosylceramidase.
Similar content being viewed by others
References
Andrews JM, Cancilla PA, Grippo J, Menkes JH (1971) Globoid cell leukodystrophy (Krabbe's disease): Morphological and biochemical studies. Neurology 21:337–352
Andrews JM, Menkes JH (1970) Ultrastructure of experimentally produced globoid cells in the rat. Exp Neurol 29:483–493
Anzil AP, Blinzinger K, Mehraein P, Dorn G, Neuhauser G (1972) Cytoplasmic inclusions in a child affected with Krabbe's disease (globoid cell leucodystrophy) and in the rabbit injected with galactocerebrosides. J Neuropathol Exp Neurol 31:370–388
Austin JH, Lehfeldt D (1965) Studies in globoid (Krabbe) leukodystrophy. III. Significance of experimentally produced globoid-like elements in rat white matter and spleen. J Neuropathol Exp Neurol 24:265–289
Blakemore WR (1972) Observation on oligodendrocyte degeneration, the resolution of status spongiosus and remyelination in Cuprizone intoxication in mice. J Neurocytol 1:413–426
D'Agostino AN, Sayre G, Hayles AB (1963) Krabbe's disease: Globoid cell type of leukodystrophy. Arch Neurol 8:82–112
Dal Canto MD, Lipton HL (1975) Primary demyelination in theiler's virus infection: An ultrastructural study. Lab Invest 33:626–637
Duchen LW, Eicher EM, Jacobs JM, Scaravilli F, Teixeira F (1980) Hereditary leucodystrophy in the mouse: The new mutant twitcher. Brain 103:695–710
Dunn HG, Dolman CL, Farrell DF, Tischler B, Hasinoff C, Woolf LI (1976) Krabbe's leukodystrophy without globoid cells. Neurology 26:1035–1041
Hodges GR, Watanabe I (1980) Chemical injury of the spinal cord of the rabbit after intracisternal injection of gentamicin. An ultrastructural study. J Neuropathol Exp Neurol 39:452–475
Karnovsky MJ (1971) Use of ferrocyanide-reduced osmium tetroxide in electron microscopy. Proc 11th Ann Meet Am Soc Cell Biol, p 146
Kobayashi T, Nagara H, Suzuki K, Suzuki K (1982) The twitcher mouse: Determination of genetic status by galactosy/ceramidase assays on clipped tail. Biochem Med 21:8–14
Kobayashi T, Yamanaka T, Jacobs JM, Teixeira F, Suzuki K (1980) The twitcher mouse: An enzymatically authentic model of human globoid cell leukodystrophy (Krabbe disease). Brain Res 202:479–483
Ludwin SK (1978) Central nervous system demyelination and remyelination in the mouse: An ultrastructural study of Cuprizone toxicity. Lab Invest 39:597–612
Miyatake T, Suzuki K (1972) Globoid cell leukodystrophy —Additional deficiency of psychosine galactosidase. Biochem Biophys Res Commun 48:538–543
Mori S, LeBlond CP (1970) Electron-microscopic identification of three classes of oligodendrocytes and a preliminary study of their proliferative activity in the corpus callosum of young rats. J Comp Neurol 139:1–29
Nagara H, Kobayashi T, Suzuki K, Suzuki K (1982) The twitcher mouse: Normal pattern of early myelination in the spinal cord. Brain Res 244:289–294
Nagara H, Suzuki K (1981) Chronological study of oligodendroglial alterations and myelination in quaking mice. Neuropathol Appl Neurobiol 7:135–149
Oehmichen M, Grüninger H (1974) Zur Entstehung von mehrkernigen Riesenzellen bei der experimentell induzierten und spontanen Krabbe'schen Krankheit (globoid cell leukodystrophy). Beitr Pathol 153:111–132
Okeda R, Suzuki Y, Horiguchi S, Fujii T (1979) Fetal globoid cell leukodystrophy in one of twins. Acta Neuropathol (Berl) 47: 151–154
Olsson R, Sourander P, Svennerholm L (1966) Experimental studies on the pathogenesis of leucodystrophies. I. The effect of intracerebrally injected sphingolipid in the rat brain. Acta Neuropathol (Berl) 6:153–163
Peters A, Palay S, Webster H deF (1976) The fine structure of the nervous system: The neurons and supporting cells. Saunders, Philadelphia, PA, pp 248–254
Powell HC, Lampert PW (1975) Oligodendrocytes and their myelinplasma membrane connections in JHM mouse hepatitis virus encephalomyelitis. Lab Invest 33:440–445
Privat A, Jacque C, Bourre JM, Dupouey P, Baumann N (1979) Absence of the major dense line in myelin of the mutant mouse, “Shiverer”. Neurosci Lett 12:107–112
Rosenbluth J (1980) Central myelin in the mouse mutant shiverer. J Comp Neurol 194:639–648
Schochet SS, Hardman JM, Lampert PW, Earle K (1969) Krabbe's disease (globoid leukodystrophy). Arch Pathol 88:305–313
Suzuki K (1970) Ultrastructural study of experimental globoid cells. Lab Invest 23:612–619
Suzuki K, De Paul L (1971) Cellular degeneration in developing central nervous system of rats produced by hypocholesteremic drug. AY9944. Lab Invest 25:546–555
Suzuki K, Grover WD (1970) Krabbe's leukodystrophy (globoid cell leukodystrophy). An ultrastructural study. Arch Neurol (Chic) 22:385–396
Suzuki K, Suzuki Y (1983) Galactosylceramide lipidosis: Globoid cell leucodystrophy (Krabbe's disease). In: Stanbury JB, Wyngaarden JB, Fredrickson DS, Goldstein JL, Brown MS (eds) The metabolic basis of inherited disease. Mc Graw-Hill, New York, pp 857–880
Suzuki K, Tanaka H, Suzuki K (1976) Studies on the pathogenesis of Krabbe's Leukodystrophy — Cellular reaction of the brain to exogenous galatosylsphingosine, monogalactosyl diglyceride and lactosylceramide, In: Volk BW, Schneck L (eds) Current trends in sphingolipidoses and allied disorders, Plenum Press, New York, pp 99–114
Vanier MT, Svennerholm L (1975) Chemical pathology of Krabbe's disease, part 3. Ceramide-hexosides and ganglioside of brain. Acta Paediat Scand 64:641–648
Wenger DA, Sattler M, Hiatt W (1974) Globoid cell leukodystrophy: Deficiency of lactosylceramide beta-galactosidase. Proc Natl Acad Sci (Warsh) 71:854–857
Wenger DA, Sattler M, Markey SP (1973) Deficiency of monogalactosyl diglyceride ß-galactosidase activity in Krabbe's disease. Biochem Biophys Res Commun 53:680–685
Yajima K (1980) Cannine globoid cell leukodystrophy: Chronological neuropathological observations in the early lesions. Brain Develop 12:153–162
Yajima K, Fletcher TF, Suzuki K (1977) Canine globoid cell leukodystrophy I. Further ultrastructural study of the typical lesion. J Neurol Sci 33:179–197
Yajima K, Suzuki K (1979) Ultrastructural changes of oligodendroglia and myelin sheaths induced by ethidium bromide. Neuropathol Appl Neurobiol 5:49–62
Yunis EJ, Lee RE (1969) The ultrastructure of globoid (Krabbe) leukodystrophy. Lab Invest 21:415–419
Yunis EJ, Lee RE (1972) Further observations on the fine structure of globoid leukodystrophy: Peripheral neuropathy and optic nerve involvement. Human Pathol 3:371–388
Author information
Authors and Affiliations
Additional information
Supported in part by research grants NS-03356, NS-10803, NS-10885, and HD-01799 and a training grant for experimental neuropathology, NS-07098, from the National Institutes of Health, USPHS, USA
Rights and permissions
About this article
Cite this article
Takahashi, H., Igisu, H. & Suzuki, K. The twitcher mouse: An ultrastructural study on the oligodendroglia. Acta Neuropathol 59, 159–166 (1983). https://doi.org/10.1007/BF00703199
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00703199