Summary
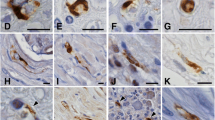
Numerous nemaline bodies were demonstrated in the pectoralis major muscle of an autopsy case of a 52-year-old woman, who died of respiratory failure after having suffered from spinal progressive muscular atrophy for 1 year. Rod-like structures ultrastructurally indistinguishable from classical nemaline bodies were abundant in both normal-appearing and atrophic myofibers of the pectoralis major muscle but not in the appendicular skeletal muscles. Morphometric analysis of spinal anterior horn cells clearly showed severe depopulation of somatic motor neurons in the lower cervical cord segments. The present case may provide further support for the neurogenic nature of nemaline body formation.
Similar content being viewed by others
References
Afifi AK, Smith JW, Zellweger H (1965) Congenital nonprogressive myopathy. Central core disease and nemaline myopathy in one family. Neurology 15:371–381
Coërs C, Telerman-Toppet N, Cremer M (1971) Regressive vacuolar myopathy in steatorrhea. Electrophysiological and histological study. Arch Neurol 24:217–227
Conen PE, Murphy EG, Donohue WL (1963) Light and electron microscopic studies of “myogranules” in a child with hypotonia and muscle weakness. Can Med Assoc J 89:983–986
Cornog JL, Gonatas NK (1967) Ultrastructure of Rhabdomyoma. J Ultrastruct Res 20:433–450
Dahl DS, Klutzow FW (1974) Congenital rod disease. Further evidence of innervational abnormalities as the basis for the clinicopathologic features. J Neurol Sci 23:371–385
Engel WK (1967) A critique of congenital myopathies and other disorders. In: Milhorat AT (ed) Exploratory concepts in muscular dystrophy and related disorders. Proceeding of an international conference, New York, 1966. International congress series 147. Excerpta Medica, Amsterdam, pp 27–40
Fardeau M (1982) Congenital myopathies. In: Mastaglia FL, Walton SJ (eds) Skeletal muscle pathology. Churchill Livingstone, Edinburgh Melbourne London New York, pp 161–203
Fukunaga H, Osame M, Igata A (1980) A case of nemaline myopathy with ophthalmoplegia and mitochondrial abnormalities. J Neurol Sci 46:169–177
Karpati G, Carpenter S, Andermann F (1971) A new concept of childhood nemaline myopathy. Arch Neurol 24:291–304
Meier C, Voellmy W, Gertsch M, Zimmermann A, Geissbühler J (1984) Nemaline myopathy appearing in adults as cardiomyopathy. A clinico-pathologic study. Arch Neurol 41:443–445
Meltzer HY, McBride E, Poppei RW (1973) Rod (nemaline) bodies in the skeletal muscle of an acute schizophrenic patient. Neurology 23:769–780
Powers JM, Young GF, Bass EB, Reed FE (1980) Atypical nemaline myopathy with temporomandibular ankylosis. Neurology 30:971–975
Rewcastle NB, Humphrey JC (1965) Vacuolar myopathy. Clinical, histochemical and microscopic study. Arch Neurol 12:570–582
Robertson WC, Kawamura Y, Dyck PJ (1978) Morphometric study of motoneurons in congenital nemaline myopathy and Werding-Hoffmann disease. Neurology 28:1057–1061
Shy GM, Engel WK, Somers JE, Wanko T (1963) Nemaline myopathy. A new congenital myopathy. Brain 86:793–810
Stoessl AJ, Hahn AF, Malott D, Jones DT, Silver MD (1985) Nemaline myopathy with associated cardiomyopathy. Report of clinical and detailed autopsy findings. Arch Neurol 42:1084–1086
Telerman-Toppet N, Gerard JM, Coërs C (1973) Central core disease. A study of clinically unaffected muscle. J Neurol Sci 19:207–223
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Konno, H., Iwasaki, Y., Yamamoto, T. et al. Nemaline bodies in spinal progressive muscular atrophy. Acta Neuropathol 74, 84–88 (1987). https://doi.org/10.1007/BF00688343
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00688343