Summary
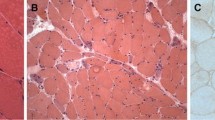
The quadriceps femoris muscles of experimental allergic myositis, in strain 13 guinea pigs immunised with rabbit myosin B fraction, were subjected to histochemical, immunohistochemical and electron microscopic studies. They demonstrated a variety of degenerative changes of muscle fibres, infiltration of lymphocytes and macrophages along with deposition of immunoglobulin G (IgG) and complement factor 3 on the surface of the muscle fibres. One third of the infiltrating cells were macrophages with acid phosphatase activity in the cytoplasm. The serum IgG of the model had an affinity for the surface of normal guinea pig muscle fibres and for thick filaments and other organelles. Its affinity for the heavy and light chains of myosin, actin, troponin T and for other proteins was shown by the immunoblotting method combined with one- and two-dimensional electrophoreses.
Similar content being viewed by others
References
Andre-Schwartz J, Datta SK, Shoenfeld Y, Isenberg DA, Stollar BD, Schwartz RS (1984) Binding of cytoskeletal proteins by monoclonal anti-DNA lupus antibodies. Clin Immunol Immunopathol 31:261–271
Barka T, Anderson PJ (1962) Histochemical methods for acid phosphatase using hexazonium pararosanilin as coupler. J Histochem Cytochem 10:741–753
Bessis M (1973) Living blood cells and their ultrastructure. Translated by Weed RI. Springer, Berlin Heidelberg New York, pp 413–476
Currie S (1971) Experimental myositis. The in vivo and in vitro activity of lymph node cells. J Pathol 105:169–185
Dawkins RL, Eghtedari A, Holborow EJ (1971) Antibodies to skeletal muscle demonstrated by immunofluorescence in experimental autoallergic myositis. Clin Exp Immunol 9:329–337
Engel AG, Arahata K (1984) Monoclonal antibody analysis of mononuclear cells in myopathies. II. Phenotypes of autoinvasive cells in polymyositis and inclusion body myositis. Ann Neurol 16:209–215
Engel AG, Biesecker G (1982) Complement activation in muscle fiber necrosis. Demonstration of the membrane attack complex of complement in necrotic fibers. Ann Neurol 12:289–296
Feltkamp TEW (1975) Technique and relevance of autoantibody determination in myasthenia gravis. In: Kunze K, Desmedt JE (eds) Studies on neuromuscular diseases. Proceedings of the International Symposium, Giessen 1973. Kargen, Basel, pp 174–179
Fulton AB (1984) The cytoskeleton. Cellular architecture and choreography. Chapman and Hall, New York, pp 33–38
Graham RC, Karnovsky MJ (1966) The early stages of absorption of injected horseradish peroxidase in the proximal tubules of mouse kidney. Ultrastructural cytochemistry by a new technique. J Histochem Cytochem 14:291–302
Karcher D, Lowenthal A, Thormar H, Noppe M (1981) Serological identification of viral antigens after electrophoretic transfer. J Immunol Methods 43:175–179
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–686
Matsubara S, Mair WGP (1979) Ultrastructural changes in polymyositis. Brain 102:701–725
Matsubara S, Takamori M (1987) Experimental allergic myositis. Strain 13 guinea pig immunized with rabbit myosin B fraction. Acta Neuropathol (Berl) (in press)
McDonald BL, Dawkins RL, Robinson J (1979) Myosin autoantibodies reacting with selective muscle fiber types. Muscle Nerve 2:37–43
McLean IW, Nakane PK (1974) Periodate-lysine-paraformaldehyde fixative. A new fixative for immuno-electron microscopy. J Histochem Cytochem 22:1077–1083
Mikawa T, Takeda S, Shimizu T, Kitaura T (1981) Gene expression of myofibrillar proteins in single muscle fibers of adult chicken. Micro two dimensional gel electrophoretic analysis. J Biochem (Tokyo) 89:1951–1962
Nishikai M, Reichlin M (1980) Heterogeneity of precipitating antibodies in polymyositis and dermatomyositis. Characterization of the Jo-1 antibody system. Arthritis Rheum 23:881–888
Oakley BR, Kirsch DR, Morris NR (1980) A simplified ultrasensitive silver stain for detecting proteins in polyacrylamide gels. Anal Biochem 105:361–363
O'Farrell PH (1975) High resolution two-dimensional electrophoresis of proteins. J Biol Chem 250:4007–4021
Ogata K, Arakawa M, Kasahara T, Shioiri-Nakano K, Hiraoka K (1983) Detection of toxoplasma membrane antigens transferred from SDS-polyacrylamide gel to nitrocellulose with monoclonal antibody and avidin-biotin, peroxidase anti-peroxidase and immunoperoxidase methods. J Immunol Methods 65:75–82
Pardo JV, Siliciano JD, Craig SW (1983) A vinculin-containing cortical lattice in skeletal muscle. Transverse lattice elements (“costameres”) mark site of attachment between myofibrils and sarcolemma. Proc Natl Acad Sci USA 80:1008–1012
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets. Procedure and some applications. Proc Natl Acad Sci USA 76:4350–4354
Author information
Authors and Affiliations
Additional information
Supported in part by a grant from the Neuroimmunological Disease Research Committee (Japanese Ministry of Health and Welfare) and by Aid for Scientific Research (Japanese Ministry of Education). This paper was read to the 13th World Congress of Neurology, Hamburg, 1985
Rights and permissions
About this article
Cite this article
Matsubara, S., Takamori, M. Experimental allergic myositis: ultrastructural, histochemical, immunological and immunohistochemical studies. Acta Neuropathol 74, 151–157 (1987). https://doi.org/10.1007/BF00692845
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00692845