Summary
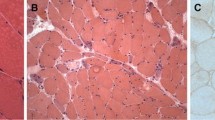
The occurrence is reported of acute myositis in a man with meningoencephalitis due to toxoplasmosis. The ultrastructure and immunohistochemistry of a muscle biopsy of the patient were investigated. Toxoplasma organisms were not found in the muscle biopsy. The perivascular inflammatory cells in the muscle were mainly CD4+ T cells and the inflammatory cells in and around the muscle fibres were chiefly macrophages. Expression of major histocompatibility complex class I and II antigens was observed in the infiltrating cells and endothelial cells of the blood vessels. A small proportion of the infiltrating cells expressed interferon-γ. A possible role of the immune mechanism in the evolution of myositis is discussed.
Similar content being viewed by others
References
Adams EM, Hafez GR, Carnes M, Wiesner JK, Graziano FM (1984) The development of polymyositis in a patient with toxoplasmosis. Clinical and pathologic findings and review of literature. Clin Exp Rheumatol 2:205–208
Appleyard ST, Dunn MJ, Dubowitz V, Rose ML (1985) Increased expression of HLA ABC class I antigens by muscle fibres in Duchenne muscular dystrophy, inflammatory myopathy, and other neuromuscular disorders. Lancet I:361–363
Bamatter F (1952) Toxoplasmosis. Mit besonderer Berücksichtigung der Embryopathia toxoplasmotica. Ergeb Inn Med Kinderheilkd 3:652–828
Behan WMH, Behan PO, Draper IT, Williams H (1983) Does toxoplasma cause polymyositis? Report of a case of polymyositis associated with toxoplasmosis and a critical review of the literature. Acta Neuropathol (Berl) 61:246–252
Chander K, Mair HJ, Mair NS (1968) Case of toxoplasma polymyositis. Br Med J 1:158–159
Emslie-Smith AM, Arahata K, Engel AG (1989) Major histocompatibility complex class I antigen expression, immunolocalization of interferon subtypes, and T cell-mediated cytotoxicity in myopathies. Hum Pathol 20:224–231
Engel AG, Arahata K (1986) Mononuclear cells in myopathies. Quantitation of functionally distinct subsets, recognition of antigen-specific cell-mediated cytotoxicity in some diseases, and implications for the pathogenesis of the different inflammatory myopathies. Hum Pathol 17:704–721
Greenlee JE, Johnson WD Jr, Campa JF, Adelman LS, Sande MA (1975) Toxoplasmosis presenting as polymyositis and cerebellar ataxia. Ann Intern Med 82:367–371
Hendrickx GFM, Verhage J, Jennekens FGI, van Knapen F (1979) Dermatomyositis and toxoplasmosis. Ann Neurol 5:393–395
Hirai N, Kobayashi A, Watanabe N, Makioka A, Hamada A, Kamio M (1988) The prevalence of toxoplasma antibodies among general outpatients and pregnant woman in Tokyo area. Jpn J Parasitol 37:261–262
Isenberg DA, Rowe D, Shearer M, Novick D, Beverley PCL (1986) Localization of interferons and interleukin 2 in polymyositis and muscular dystrophy. Clin Exp Immunol 63:450–458
Jackson MH, Hutchison WM, Siim JC (1987) A seroepidemiological survey of toxoplasmosis in Scotland and England. Ann Trop Med Parasitol 81:359–365
Karasawa T, Takizawa I, Morita K, Ishibashi H, Kanayama S, Shikata T (1981) Polymyositis and toxoplasmosis. Acta Pathol Jpn 31:675–680
Karpati G, Pouliot Y, Carpenter S (1988) Expression of immunoreactive major histocompatibility complex products in human skeletal muscles. Ann Neurol 23:64–72
Kass EH, Andrus SB, Adams RD, Turner FC, Feldman HA (1952) Toxoplasmosis in the human adult. Arch Intern Med 89:759–782
Kayhoe DE, Jacobs L, Beye HK, McCullough NB (1957) Acquired toxoplasmosis. Observations on two parasitologically proved cases treated with pyrimethamine and triple sulfonamides. N Engl J Med 257:1247–1254
Layzer RB (1985) Neuromuscular manifestations of systemic disease. F. A. Davis, Philadelphia, pp 170–173
Matsubara S, Mair WGP (1979) Ultrastructural changes in polymyositis. Brain 102:701–725
Matsubara S, Takamori M (1987) Experimental allergic myositis. Ultrastructural, histochemical, immunological and immunohistochemical studies. Acta Neuropathol (Berl) 74:151–157
McNicholl B, Underhill D (1970) Toxoplasmic polymyositis. Irish J Med Sci 3:525–527
Quilis MR, Damjanov I (1982) Dermatomyositis as an immunological complication of toxoplasmosis. Acta Neuropathol (Berl) 58:183–186
Remington JS, Krahenbuhl JL, Mendenhall JW (1972) A role for activated macrophages in resistance to infection with toxoplasma. Infect Immun 6:829–834
Rosenberg NL, Kotzin BL (1989) Abberant expression of class II MHC antigens by skeletal muscle endothelial cells in experimental autoimmune myositis. J Immunol 141:4289–4294
Rowland LP, Greer M (1961) Toxoplasmic polymyositis. Neurology 11:367–370
Samuels BS, Rietschel RL (1976) Polymyositis and toxoplasmosis. J Am Med Assoc 235:60–61
Schröter HM, Sarnat HB, Matheson DS, Selad TP (1987) Juvenile dermatomyositis induced by toxoplasmosis. J Child Neurol 2:101–104
Suzuki Y, Conley FK, Remington JS (1989) Importance of endogenous IFN-γ for prevention of toxoplasmic encephalitis in mice. J Immunol 143:2045–2050
Syverton JT, Slavin HB (1946) Human toxoplasmosis. J Am Med Assoc 131:957–959
Topi GC, D'Alessandro L, Catricala C, Zardi O (1979) Dermatomyositis-like syndrome due to toxoplasma. Br J Dermatol 101:589–591
Zuelzer WW (1944) Infantile toxoplasmosis with a report of three new cases, including two in which the patients were identical twins. Arch Pathol 38:1–19
Zuk JA, Fletcher A (1988) Skeletal muscle expression of class II histocompatibility antigens (HLA-DR) in polymyositis and other muscle disorders with an inflammatory infiltrate. J Clin Pathol 41:410–414
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Matsubara, S., Takamori, M., Adachi, H. et al. Acute toxoplasma myositis: an immunohistochemical and ultrastructural study. Acta Neuropathol 81, 223–227 (1990). https://doi.org/10.1007/BF00334513
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00334513