Summary
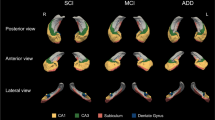
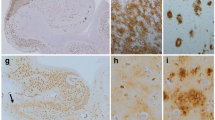
The occurrence and topographic analysis of granulovacuolar degeneration (GVD) in the hippocampal cortex of mentally normal controls (75 cases) and patients with Alzheimer's dementia (AD; 17 cases which included Alzheimer's disease and senile dementia of Alzheimer type), multi-infarct dementia (MID; 16 cases), Pick's disease (PD; 5 cases) and atypical dementia [5 cases; non-Alzheimer, non-Pick dementia with Fahr's syndrome (NANPDF)] were investigated. GVD was rarely found in control cases below the age of 60 years. In elderly normal brains, the statistically most representative ranking order of predilection for GVD (in decreasing severity) was: in the 60 s, CA1>prosubiculum >CA2 (no GVD was found in the CA3 and CA4); in the 70 s, CA1>prosubiculum >CA2 >CA3>CA4; in the 80 s, CA1>prosubiculum >CA2>CA3>CA4; in the 90s, CA1>prosubiculum >CA2>CA3>CA4. In the brains of demented patients, the rank order for GVD was: for AD, CA1 >CA2>CA3> prosubiculum >CA4; for MID, CA1 > prosubiculum >CA2>CA3>CA4; for PD, CA1 >CA2>CA3> prosubiculum >CA4; and for atypical dementia (NANPDF), CA1>CA2> prosubiculum >CA3>CA4. The similarity of the predilection to ranking order was noted both in normal aged subjects and in MID as well as both in AD and in PD. The qualitative investigation disclosed that the affected neurons with GVD in the cases of AD were found in all the examined areas outside the hippocampus (gyrus praecentralis, temporal and occipital cortex, globus pallidus, amygdaloid nuclei, mammillary bodies, medial thalamic nuclei, red nuclei, nuclei basalis of Meynert, dentate nuclei and inferior olivary nuclei) and, contrary to the results in AD, there were no affected neurons with GVD in the cases of MID. The occurrence and distribution of GVD in demented patients were different in these respective disorders.
Similar content being viewed by others
References
Ball MJ (1977) Neuronal loss, neurofibrillary tangles and granulovacuolar degeneration in the hippocampus with ageing and dementia: a quantitative study. Acta Neuropathol (Berl) 37:111–118
Ball MJ (1978) Topographic distribution of neurofibrillary tangles and granulovacuolar degeneration in hippocampal cortex of aging and demented patients: a quantitative study. Acta Neuropathol (Berl) 42:73–80
Ball MJ, Lo P (1977) Granulovacuolar degeneration in the aging brain and in dementia. J Neuroapthol Exp Neurol 36:474–487
Ball MJ, Nuttall K (1981) Topography of neurofibrillary tangles and granulovacuoles in hippocampi of patients with Down's syndrome: quantitative comparison with normal ageing and Alzheimer's disease. Neuropathol Applied Neurobiol 7:13–20
Ball MJ, Vis CL (1978) Relationship of granulovacuolar degeneration in hippocampal neurons to aging and to dementia in normal-pressure hydrocephalics. J Gerontol 6:815–824
Bondareff W, Wischik CM, Novak M, Roth M (1991) Sequestration of Tau by granulovacuolar degeneration in Alzheimer's disease. Am J Pathol 130:641–647
Dickson DW, Ksiezak-Reding H, Davies P, Yen S-H (1987) A monoclonal antibody that recognizes a phosphorylated epitope in Alzheimer neurofibrillary tangles, neurofilaments and tau proteins immunostains granulovacuolar degeneration. Acta Neuropathol (Berl) 73:254–258
Hirano A, Dembitzer HM, Kurland LT (1968) The fine structure of some intraganglionic alterations. Neurofibrillary tangles, granulovacuolar bodies and “rod-like” structures as seen in Guam amyotrophic lateral sclerosis and Parkinson's-dementia complex. J Neuropathol Exp Neurol 67:167–182
Kahn J, Anderton BH, Probst A, Ulrich J, Esiri MM (1985) Immunohistological study of granulovacuolar degeneration using monoclonal antibodies to neurofilaments. J Neurol Neurosurg Psychiatry 48:924–926
Lorente de No R (1934) Studies on the structure of the cerebral cortex. II. Continuation of the study of the ammonic system. J Psychol Neurol 46:113–177
Mena R, Robitaille Y, Cuello AC (1990) Granulovacuolar degeneration cores cross-react with a tau-microtubular mAb of high specificity for PHFs. J Neuropathol Exp Neurol 49:308
Shibayama H, Kobayashi H, Iwase S, Nakagawa M, Marui Y, Kayukawa Y, Iwata H, Takeuchi T (1986) Unusual cases of presenile dementia with Fahr's syndrome. Jpn J Psychiatr Neurol 40:85–100
Shibayama H, Kobayashi H, Nakagawa M, Yamada K, Iwata H, Iwai K, Takeuchi T, Xu M, Ishihara R, Kitoh J (1991) Non-Alzheimer non-Pick dementia with Fahr's syndrome (NANPDF). Clin Neuropathol (in press)
Simchowicz T (1911) Histopathologische Studien über die senile Demenz. In: Nissl F, Alzheimer A (eds) Histologie und histopathologische Arbeiten. Über die Großhirnrinde, vol 4. Fischer, Jena, pp267–444
Tomlinson BE, Blessed G, Roth M (1968) Observations on the brains of non-demented old people. J Neurol Sci 7:331–356
Tomlinson BE, Blessed G, Roth M (1970) Observations on the brains of demented old people. J Neurol Sci 11:205–242
Tomlinson BE, Kitchener D (1972) Granulovacuolar degeneration of hippocampal pyaramidal cells. J Pathol 106:165–185
Woodard JS (1962) Clinicopathologic significance of granulovacuolar degeneration in Alzheimer's disease. J Neuropathol Exp Neurol 21:85–91
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Xu, M., Shibayama, H., Kobayashi, H. et al. Granulovacuolar degeneration in the hippocampal cortex of aging and demented patients — a quantitative study. Acta Neuropathol 85, 1–9 (1992). https://doi.org/10.1007/BF00304627
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00304627