Abstract
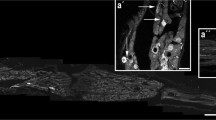
Calcitonin gene-related peptide (CGRP) and substance P (SP)-immunoreactive (IR) axons in the nucleus gracilis of normal rats (1–15 months of age) were studied by light and electron microscopy. Besides many CGRP-IR and SP-IR varicosities with normal appearance, we found a few swollen (nearly round or oval) varicosities with either CGRP or SP immunoreactivity. Swollen CGRP-IR varicosities were more frequently seen than SP-IR ones, appearing from 3 months of age and increasing in number and size (up to approximately 25 μm in diameter) with advancing age. At the electron microscopic (EM) level, CGRP-IR and SP-IR swollen varicosities showed dystrophic changes, i.e., many membranous dense bodies, and proliferation of microtubules and neurofilaments. CGRP-IR or SP-IR dystrophic axons also contained many mitochondria and sometimes made synaptic contacts with nonreactive dendrites (occasionally with non-IR axons). These findings suggest that the dystrophic CGRP and SP axonal profiles represent a functionally distinct subpopulation of axonal dystrophy in the nucleus gracilis and use CGRP or SP as a neuroactive substance. Using a double-immunostaining method, many of normal CGRP-IR axons were identified to be SP-IR. However, no single dystrophic varicosity was found to contain both CGRP and SP immunoreactivities. These findings suggest that CGRP and SP afferents are independently affected and progress to dystrophic changes.
Similar content being viewed by others
References
Biella G, Panara C, Pecile A, Sotgiu ML (1991) Facilitatory role of calcitonin gene-related peptide (CGRP) on excitation induced by substance P (SP) and noxious stimuli in rat spinal dorsal horn neurons: an iontophoretic study in vivo. Brain Res 559:352–356
Conti F, De Biasi S, Giuffrida R, Rustioni A (1990) Substance P-containing projections in the dorsal columns of rats and cats. Neuroscience 34:607–621
Farmer PM, Wisniewski HW, Terry RD (1976) Origin of dystrophic axons in the gracile nucleus. J Neuropathol Exp Neurol 35:366
Fujisawa K (1988) Study of axonal dystrophy. III. Posterior funiculus and posterior column of ageing and old rats. Acta Neuropathol 76:115–127
Fujisawa K, Shiraki H (1980) Study of axonal dystrophy. II. Dystrophy and atrophy of the presynaptic boutons: a dual pathology. Neuropathol Appl Neurobiol 6:387–398
Inagaki S, Kubota Y, Kito S, Yamano M, Tohyama M (1986) Enkephalin-like immunoreactive neurons and fibers in the ventromedial hypothalamic nucleus. Brain Res Bull 17:159–167
Ju G, Hökfelt T, Brodin E, Fahrenkrug J, Fisher JA, Frey P, Elde RP, Brown JC (1987) Primary sensory neurons of the rat showing calcitonin gene-related peptide immunoreactivity and their relation to substance P-, somatostatin,, galanin-, vasoactive intestinal polypeptide- and cholecystokinin-immunoreactive ganglion cells. Cell Tissue Res 247:417–431
Katayama-Kumoi Y, Kiyama H, Manabe R, Shiotani Y, Tohyama M (1985) Coexistence of glucagon- and substance P-like immunoreactivity in the chicken retina. Neuroscience 16: 417–424
Kikuchi T, Mukoyama M, Yamazaki K, Moriya H (1990) Axonal degeneration of ascending sensory neurons in gracile axonal dystrophy mutant mouse. Acta Neuropathol 80:145–151
Krnjevic K, Morris ME (1974) An excitatory action of substance P on cuneate neurons. Can J Physiol Pharmacol 52: 736–744
Kruger L, Sternini C, Brecha NC, Mantyh PW (1988) Distribution of calcitonin gene-related peptide immunoreactivity in relation to the rat central somatosensory projection. J Comp Neurol 273:149–162
Lee Y, Takami K, Kawai Y, Girgis S, Hillyard CJ, MacIntyre I, Emson PC, Tohyama M (1985) Distribution of calcitonin gene-related peptide in the rat peripheral nervous system with reference to its coexistence with substance P. Neuroscience 15: 1227–1237
Lundberg JM, Franco-Cereceda A, Hua X, Hökfelt T, Fischer JA (1985) Co-existence of substance P and calcitonin gene-related peptide-like immunoreactivities in sensory nerves in relation to cardiovascular and bronchoconstrictor effects of capsaicin. Eur J Pharmacol 108:315–319
Matsuda T, Maeda M, Morishima Y, Hashimoto S, Tateishi K, Hamaoka T, Mizuta H, Takagi H (1985) Dystrophic axons in the nucleus gracilis of the normal rat containing cholecystokinin-like immunoreactivity. Light- and electron-microscopic observations. Acta Neuropathol (Berl) 65:224–234
Morishima Y, Takagi H, Kawai Y, Emson PC, Hillyard CJ, Girgis SI, MacIntyre I (1986) Ultrastructural observation of nerve fibers containing both substance P and calcitonin generelated peptide in the nucleus tractus solitarii of the rat: a combination of immunofluorescence and PAP methods. Brain Res 379:157–161
Morris HR, Panico M, Etienne T, Tippins J, Girgis SI, MacIntyre I (1984) Isolation and characterization of human alcitonin gene-related peptide. Nature 308:746–748
Pentschew A, Schwarz K (1962) Systemic axonal dystrophy in vitamin-E deficient adult rats. Acta Neuropathol (Berl) 1: 313–334
Pillai SR, Traber MG, Kayden HJ, Cox NR, Toivio-Kinnucan M, Wright JC, Braund KG, Whitley RD, Gilger BC, Steiss JE (1994) Concomitant brainstem axonal dystrophy and necrotizing myopathy in vitamin E-deficient rats. J Neurol Sci 123: 64–73
Schmidt RE, Coleman BD, Nelson JS (1991) Differential effect of chronic vitamin E deficiency on the development of neuroaxonal dystrophy in rat gracile/cuneate nuclei and prevertebral sympathetic ganglia. Neurosci Lett 123:102–106
Seitelberger F (1986) Neuroaxonal dystrophy: its relation to aging and neurological diseases. Handb Clin Neurol 5: 391–415
Shimada S, Inagaki S, Kubota Y, Kito S, Shiotani Y, Tohyama M (1988) Coexistence of substance P and neurotensin-like peptides in single neurons of the rat hypothalamus. Peptides 9: 71–76
Somogyi P, Takagi H (1982) A note on the use of picric acidparaformaldehyde-glutaraldehyde fixative for correlated light-and electron-microscopic immunocytochemistry. Neuroscience 7:1779–1783
Sotelo C, Palay SL (1971) Altered axons and axon terminals in the lateral vestibular nucleus of the rat. Possible examples of axonal remodeling. Lab Invest 25:653–671
Sternberger LA (1979) Immunocytochemistry, 2nd edn. Wiley, New York
Takeuchi IK, Aoki E, Takeuchi YK (1994) Age-related and region-specific increase in number of concentric lamellar bodies in axon terminals and presynaptic axons in central nervous system of groggy mutant rat. Acta Neuropathol 87:628–634
Tamatani M, Senba E, Tohyama M (1989) Calcitonin gene-related peptide- and substance P-containing primary afferent fibers in the dorsal column of the rat. Brain Res 495:122–130
Tay SSW, Wong WC (1991) Gracile nucleus of streptozotocin-induced diabetic rats. J Neurocytol 20:356–364
Walkley SU, Baker HJ, Rattazzi MC, Haskins ME, Wu JY (1991) Neuroaxonal dystrophy in neuronal storage disorders: evidence for major GABAergic neuron involvement. J Neurol Sci 104:1–8
Woolf CJ, Wiesenfeld-Hallin Z (1986) Substance P and calcitonin gene-related peptide synergistically modulate the gain of the nociceptive flexor withdrawal reflex in the rat. Neurosci Lett 66:226–230
Yoshikawa H, Tarui S, Hashimoto PH (1985) Diminished retrograde transport causes axonal dystrophy in the nucleus gracilis. Acta Neuropathol (Berl) 68:93–100
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fujiwara, K., Baek, S.Y., Arakawa, T. et al. Calcitonin gene-related peptide- and substance P-immunoreactive axons in the nucleus gracilis of the rat with special reference to axonal dystrophy: light and electron microscopic observations. Acta Neuropathol 90, 347–355 (1995). https://doi.org/10.1007/BF00315008
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00315008