Summary
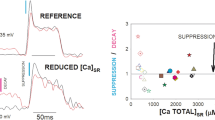
Depolarization-induced Ca2+ release was studied in a mixture of triads and terminal cisternae isolated from rabbit skeletal muscle. The vesicles were actively loaded with known amounts of Ca2+ in the absence of precipitating anions in a solution containing 100 mm K propionate buffer. Changes in extravesicular Ca2+ were monitored with 10 μ m Fura-2 (membrane impermeant form). Ca2+ release was initiated by diluting an aliquot of the loaded vesicles into a TEACl release solution designed to maintain a constant [K+] · [Cl−] product. Fast release, defined as the percentage of total Ca2+ loaded which released in less than 10 sec, occurred when extravesicular free Ca2+ was in the submicromolar range and was unaffected by 5 mm caffeine under depolarizing conditions, change in external pH to 6.5, and an increase in external Mg2+ concentration from 0.1 to 0.2 mm. Thus, the Ca2+ release measured in these studies is distinct from Ca2+-induced Ca2+ release. The fast release more than doubled when a greater dilution (1 ∶ 20 versus 1 ∶ 10) of the loaded vesicles into the release solution, which would produce a larger depolarization, was used. The percentage of loaded Ca2+ which released rapidly in a particular triad preparation was similar to the percentage of vesicles structurally coupled as visualized by electron microscopy.
Similar content being viewed by others
References
Beeler, T., Russell, J.T., Martonosi, A. 1979. Optical probe responses on sarcoplasmic reticulum: Oxacarbocyanines as probes of membrane potential. Eur. J. Biochem. 95:579–591
Bradford, M.M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principles of protein dye binding. Anal. Biochem. 72:248–254
Caswell, A.H., Lau, Y.H., Brunschwig, J.-P. 1976. Ouabainbinding vesicles from skeletal muscle. Arch. Biochem. Biophys. 176:417–430
Donaldson, S.K.B. 1985. Peeled mammalian skeletal muscle fibers: Possible stimulation of Ca2+ release via a transverse tubule-sarcoplasmic reticulum mechanism. J. Gen. Physiol. 86:501–525
Endo, M. 1975. Mechanism of action of caffeine on the sarcoplasmic reticulum of skeletal muscle. Proc. Jpn. Acad. 51:479–484
Grynkiewicz, G., Poenie, M., Tsien, R. 1985. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J. Biol. Chem. 260:3440–3450
Hildalgo, C., Gonzalez, M.E., Lagos, R. 1983. Characterization of the Ca2+or Mg2+-ATPase of transverse tubule membranes isolated from rabbit skeletal muscle. J. Biol. Chem. 258:13937–13945
Ikemoto, N., Antoniu, B., Kim, D.H. 1984. Rapid calcium release from the isolated sarcoplasmic reticulum is triggered via the attached transverse tubular system. J. Biol. Chem. 259:13151–13158
Ikemoto, N., Antoniu, B., Mészáros, L.G. 1985. Rapid flow chemical quench studies of calcium release from isolated sarcoplasmic reticulum. J. Biol. Chem. 260:14096–14100
Imagawa, T., Smith, J., Coronado, R., Campbell, K. 1987. Purified ryanodine receptor from skeletal muscle sarcoplasmic reticulum is the Ca2+-permeable pore of the calcium release channel. J. Biol. Chem. 262:16636–16643
Kasai, M., Miyamoto, H. 1973. Depolarization induced calcium release from sarcoplasmic reticulum membrane fragments by changing ionic environment. FEBS Lett. 34:299–301
Kasai, M., Miyamoto, H. 1976a. Depolarization induced calcium release from sarcoplasmic reticulum fragments: I. Release of calcium taken up upon using ATP. J. Biochem. 79:1053–1066
Kasai, M., Miyamoto, H. 1976b. Depolarization induced calcium release from sarcoplasmic reticulum fragments: II. Release of calcium incorporated without ATP. J. Biochem. 79:1067–1076
Kim, D.H., Ohnishi, S.T., Ikemoto, N. 1983. Kinetic studies of calcium release from sarcoplasmic reticulum. J. Biol. Chem. 258:9662–9668
Lau, Y.H., Caswell, A.H., Brunschwig, J.-P. 1977. Isolation of transverse tubules by fractionation of triad junctions of skeletal muscle. J. Biol. Chem. 252:5565–5574
Lowry, O.H., Rosebrough, N.J., Farr, A.L., Randall, R.J. 1951. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193:265–276
MacLennan, D.H., Shoshan, V., Wood, D.S. 1983. Studies of Ca2+ release from sarcoplasmic reticulum. Ann. N.Y. Acad. Sci. 402:470–476
Martonosi, A., Feretos, R. 1964. Sarcoplasmic reticulum: I. The Uptake of Ca++ by sarcoplasmic reticulum fragments. J. Biol. Chem. 239:648–658
Meissner, G. 1984. Adenine nucleotide stimulation of Ca2+ release in sarcoplasmic reticulum. J. Biol. Chem. 259:2365–2374
Meissner, G. 1986. Evidence of a role for calmodulin in the regulation of calcium release from skeletal muscle sarcoplasmic reticulum. Biochemistry 25:244–251
Meissner, G., Darling, E., Eveleth, J. 1986. Kinetics of rapid Ca2+ release by sarcoplasmic reticulum. Effects of Ca2+, Mg2+, and adenine nucleotides. Biochemistry 25:236–244
Meissner, G., Henderson, J.S. 1987. Rapid calcium release from cardiac sarcoplasmic reticulum vesicles is dependent on Ca2+ and is modulated by Mg2+, adenine nucleotide and calmodulin. J. Biol. Chem. 262:3065–3073
Meissner, G., McKinley, D. 1976. Permeability of sarcoplasmic reticulum membrane: The effect of changed ionic environments on Ca2+ release. J. Membrane Biol. 30:79–98
Melzer, W., Schneider, M.F., Simon, B.J., Szücs, G. 1986. Intramembrane charge movement and calcium release in frog skeletal muscle. J. Physiol. 373:481–511
Mitchell, R.D., Palade, P., Fleischer, S. 1983. Purification of morphologically intact triad structures from skeletal muscle. J. Cell Biol. 96:1008–1016
Miyamoto, H., Racker, E. 1982. Mechanism of calcium release from skeletal sarcoplasmic reticulum. J. Membrane Biol. 66:193–201
Mobley, B.A. 1979. Chloride and osmotic contractures in skinned frog muscle fibers. J. Membrane Biol. 46:315–329
Ohkusa, T., Carlos, A.D., Kang, J.J., Smilowitz, H., Ikemoto, N. 1991. Effects of dihydropyridines on calcium release from the isolated membrane complex consisting of the transverse tubule and sarcoplasmic reticulum. Biochem. Biophys. Res. Commun. 175:271–276
Ohkusa, T., Smilowitz, H.M., Ikemoto, N. 1990. Effects of the dihydropyridine (DHP) receptor-specific agents on depolarization-indued Ca2+ release from SR in vitro. Biophys. J. 57:498a
Palade, P. 1987. Drug-induced Ca2+ release from isolated sarcoplasmic reticulum: II. Releases involving a Ca2+-induced Ca2+ release channel. J. Biol. Chem. 262:6142–6148
Palade, P., Mitchell, R.D., Fleischer, S. 1983. Spontaneous calcium release from sarcoplasmic reticulum: General description and effects of calcium. J. Biol. Chem. 258:8098–8107
Rios, E., Brum, G. 1987. Involvement of dihydropyridine receptors in excitation-contraction coupling in skeletal muscle. Nature 325:717–720
Rousseau, E., LaDine, J., Liu, Q.Y., Meissner, G. 1988. Activation of the Ca2+ release channel of skeletal muscle sarcoplasmic reticulum by caffeine and related compounds. Arch. Biochem. Biophys. 267:75–86
Rousseau, E., Pinkos, J. 1990. pH modulates conducting and gating behaviour of single calcium release channels. Pfluegers Arch. 415:645–647
Stephenson, E.W. 1985a. Excitation of skinned muscle fibers by imposed ion gradients: I. Stimulation of 45Ca efflux at constant [K][Cl] product. J. Gen. Physiol. 86:813–832
Stephenson, E.W. 1985b. Excitation of skinned muscle fibers by imposed ion gradients: II. Influence of quercetin and ATP removal on the Ca2+-insensitive component of stimulated 45Ca efflux. J. Gen. Physiol. 86:833–852
Volpe, P., Stephenson, E.W. 1986. Ca2+ dependence of transverse-tubule mediated calcium release in skinned skeletal muscle fibers. J. Gen. Physiol. 87:271–288
Author information
Authors and Affiliations
Additional information
We thank Gerry Vaio and Melanie Vander Klok for excellent technical support. This work was supported by the National Institutes of Health program project grant PO1-HL27867, NSF Biological Instrumentation Grant DIR-8812094 and State of Ohio Research Challenge Grant.
Rights and permissions
About this article
Cite this article
Corbett, A.M., Bian, J., Wade, J.B. et al. Depolarization-induced calcium release from isolated triads measured with impermeant Fura-2. J. Membarin Biol. 128, 165–179 (1992). https://doi.org/10.1007/BF00231810
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00231810