Abstract
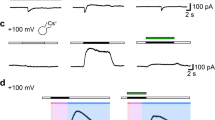
Using the inside-out patch clamp technique, we identified a Cl− channel in patches from the membrane of cultured human hematopoietic myeloblastic leukemia ML-1 cells. The Cl− channel was not seen at negative membrane potentials in excised patches until the membrane potential was depolarized to greater than +40 mV. The channel was also activated by addition of cAMP-dependent protein kinase (PKA) catalytic subunit at physiological membrane potential (−40 mV). Biophysical studies of the Cl− channel revealed that the current-voltage (I-V) relationship of the Cl− channel was outwardly rectifying in symmetrical 142 mm Cl− solutions. Single channel conductances were 48 pS for the outward current measured at +60 mV and 27 pS for the inward current at −60 mV. The open time constant of the channel was dependent on the membrane potential and was significantly prolonged at positive membrane potentials. Channels activated by cAMP-dependent protein kinase spent a significantly longer time in the open state compared to those channels activated by depolarization pulses. Pharmacological properties of the Cl− channel were also studied. Two anion transport inhibitors, anthracene-9-carboxylic acid (9-AC) and 4,4-diisothiocyanatostilbene-2,2-disulfonic acid (DIDS) caused a flickering block of the channel. Half-inhibitory concentrations (IC50) for 9-AC and DIDS were 174 ± 20 and 70±16 μm, respectively. Blockade of the Cl− channel by 9-AC or DIDS was completely reversible. Our findings suggest that outwardly rectifying Cl− channels (ORCC) are present in human hematopoietic myeloblasts. The function of ORCC may be involved in hormone-regulated cell growth, cell volume regulation and immune responses.
Similar content being viewed by others
References
Bear, C.E. 1988. Phosphorylation-activated chloride channels in human skin fibroblasts. FEBS Lett. 237:145–149
Berger, H.A., Anderson, M.P., Gregory, R.J., Thompson, S., Howard, P.W., Maurer, R.A., Mulligan, R., Smith, A.E., Welsh, M.J. 1991. Identification and regulation of the cystic fibrosis transmembrane conductance regulator-generated chloride channel. J. Clin. Invest. 88:1422–1431
Bridges, R.J., Worrel, R.T., Frizzell, R.A., Benos, D.J. 1989. Stilbene disulfonate blockade of colonic secretory Cl− channels in planar bilayers. Am. J. Physiol. 256:C902-C912
Bubien, J.K., Kirk, K.L., Rado, T.A., Frizzell, R.A. 1990. Cell cycle dependence of chloride permeability in normal and cystic fibrosis lymphocytes. Science 248:1416–1419
Chen, J.H., Schulman, H., Gardner, P. 1989. A cAMP-regulated chloride channel in lymphocytes that is affected in cystic fibrosis. Science 243:657–660
Craig, R.W., Frankfurt, O.S., Sakagami, H., Takeda, K., Bloch, A. 1984. Macromolecular and cell cycle effects of different classes of agents inducing the maturation of human myeloblastic leukemia (ML-1) cells. Cancer Res. 44:2421–2429
Egan, M., Flotte, T., Aflone, S., Solow, R., Zeitlin, P.L., Carter, B.J., Guggino, W.B. 1992. Defective regulation of outwardly rectifying Cl− channels by protein kinase A corrected by insertion of CFTR. Nature 358:581–584
Frizzell, R.A., Rechkemmer, G., Shoemaker, R.L. 1986. Altered regulation of airway epithelial cell chloride channels in cystic fibrosis. Science 233:558–560
Gabriel, S.E., Clarke, L.L., Boucher, R.C., Stutts, M.J. 1993. CFTR and outward rectifying chloride channels are distinct proteins with a regulatory relationship. Nature 363:263–266
Garber, S.S. 1992. Outwardly rectifying chloride channels in lymphocytes. J. Membrane Biol. 127:49–56
Gogelein, H. 1988. Chloride channels in epithelia. Biochim. Biophys. Acta 947:521–547
Gray, M.A., Harris, A., Coleman, L., Greenwell, J.R., Argent, B.E. 1989. Two types of chloride channel on duct cells cultured from human fetal pancreas. Am. J. Physiol. 26:C240-C251
Halm, D.R., Rechkemmer, G.R., Schoumacher, R.A., Frizzell, R.A. 1988. Apical membrane chloride channels in a colonie cell line activated by secretory agonists. Am. J. Physiol. 254:C505-C511
Hanrahan, J.W., Tabcharani. 1990. Inhibition of outwardly rectifying anion channel by HEPES and related buffers. J. Membrane Biol 116:65–70
Hwang, T.-C., Guggino, S.E., Guggino, W.B. 1990. Direct modulation of secretory chloride channels by arachidonic and other cis unsaturated fatty acids. Proc. Natl. Acad. Sci. USA 87:5706–5709
Hwang, T.-C., Lu, L., Zeitlin, P.L., Gruenert, D.C., Huganir, R., Guggino, W.B. 1989. Cl− channels in CF: lack of activation by protein kinase C and cAMP-dependent protein kinase. Science 244:1351–1353
Li, M., McCann, J.D., Anderson, M.P., Clancy, J.P., Liedtke, C.M., Nairn, A.C., Greengard, P., Welsh, M.J. 1989. Regulation of chloride channels by protein kinase C in normal and cystic fibrosis airway epithelia. Science 244:1353–1356
Li, M., McCann, J.D., Liedtke, C.M., Nairn, A.C., Greengard, P., Welsh, M.J. 1988. Cyclic AMP-dependent protein kinase opens chloride channels in normal but not cystic fibrosis airway epithelium. Nature 331:358–360
Lu, L., Markakis, D., Gsggmar, W.B. 1991. Blockade of Ca2+-dependent Cl− current in Xenopus oocytes by Cl− channel blockers and antimalarial drugs. Cell Physiol. Biochem, 1:251–262
Lu, L., Markakis, D., Guggino, W.B. 1993a. Identification and regulation of whole-cell Cl− and Ca2+-activated K+ currents in cultured medullary thick ascending limb cells. J. Membrane Biol. 135:181–189
Lu, L., Yang, T., Markakis, D., Guggino, W.B., Craig, R.W. 1993b. Alteration in a voltage-gated K+ current during the differentiation of ML-1 human myeloblastic leukemia cells. J. Membrane Biol. 132:267–274
Lukacs, G.L., Moczydlowski, E. 1990. A chloride channel from lobster walking leg nerves: characterization of single-channel properties in planar bilayers. J. Gen. Physiol. 96:707–733
Mauro, T.M., Isseroff, R.R., Lasarow, R., Pappone, P.A. 1993. Ion channels are linked to differentiation in keratinocytes. J. Membrane Biol. 132:201–209
Paulmichl, M., Li, Y., Wickman, K., Ackerman, M., Peralta, E., Clapham, D. 1992. New mammalian chloride channel identified by expression cloning. Nature 356:238–241
Perozo, E., Bezanilla, F. 1990. Phosphorylation affects voltage gating of the delayed rectifier K+ channel by electrostatic interaction. Neuron 5:686–690
Rafizadeh, C.M., Guggino, W.B., Montrose, M.H. 1991. Cellular differentiation regulates expression of Cl− transport and cystic fibrosis transmembrane conductance regulator mRNA in human intestinal cells. J. Biol. Chem. 266:4495–4499
Schoumacher, R.A., Shoemaker, R.L., Halm, D.R., Tallant, E.A., Wallace, R.W., Frizzell, R.A. 1987. Phosphorylation fails to activate chloride channels from cystic fibrosis airway cells. Nature 330:752–754
Singh, A.K., Afink, G.B., Venglarik, C.J. 1991. Colonie Cl− channel blockade by three classes of compounds. Am. J. Physiol. 260:C51-C63
Smith, J.J., Welsh, M.J. 1992. cAMP stimulates bicarbonate secretion across normal but not cystic fibrosis airway epithelia. J. Clin. Invest. 89:1148–1153
Solc, C.K., Wine, J.J. 1991. Swelling-induced and depolarization-induced Cl− channels in normal and cystic fibrosis epithelial cells. Am. J. Physiol. 261:C658-C674
Tabcharani, J.A., Hanrahan, J.W. 1991. On the activation of outwardly rectifying anion channels in excised patches. Am. J. Physiol. 261:G992-G999
Tabcharani, J.A., Jensen, T.J., Riordant, J.R., Hanrahan, J.W. 1989. Bicarbonate permeability of the outwardly rectifying anion channel. J. Membrane Biol. 112:109–122
Tilmann, M., Kunzelmann, K., Frobe, U., Cabantchik, I., Lang, H.J., Englert, H.C., Greger, R. 1991. Different types of blockers of the intermediate-conductance outwardly rectifying chloride channel in epithelia. Pfluegers Arch. 418:556–563
Vaca, L., Kunze, D.L. 1993. cAMP-dependent phosphorylation modulates voltage gating in an endothelial Cl− channel. Am. J. Physiol. 264:C370-C375
Venglarik, C.J., Singh, A.K., Wang, R., Bridges, R.J. 1993. Trinitrophenyl-ATP blocks colonic Cl− channels in planar phospholipid bilayers. J. Gen. Physiol. 101:545–569
Welsh, M.J. 1987. Electrolyte transport by airway epithelia. Physiol. Rev. 67:1143–1184
Welsh, M.J., Li, M., McCann, J.D. 1989. Activation of normal and cystic fibrosis Cl− channels by voltage, temperature, and trypsin. J. Clin. Invest. 84:2002–2007
Wieland, S.J., Chou, R.H., Chen, T.A. 1987. Evaluation of a potassium current in differentiating human leukemia (HL-60) cells. J. Cell. Physiol. 132:371–375
Willumsen, N.J., Boucher, R.C. 1989. Activation of an apical Cl− conductance by Ca2+ ionophores in cystic fibrosis airway epithelia. Am. J. Physiol. 256:C226-C233
Worrell, R.T., Butt, A.G., Cliff, W.H., Frizzell, R.A. 1989. A volume-sensitive chloride conductance in human colonic cell line T84. Am. J. Physiol. 256:C1111-C1119
Author information
Authors and Affiliations
Additional information
We thank Dr. R.E. White and Ms. M.P. Nardino for reviewing the manuscript. This study was partly supported by National Institutes of Health grant GM46834 (to L.L.).
Rights and permissions
About this article
Cite this article
Xu, B., Lu, L. Protein kinase A-regulated Cl− channel in ML-1 human hematopoietic myeloblasts. J. Membarin Biol. 142, 65–75 (1994). https://doi.org/10.1007/BF00233384
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00233384