Abstract
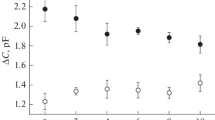
We examined the effects of seven n-alkyl alcohols (from n-butyl to n-undecyl alcohol), isoamyl alcohol and benzyl alcohol on the activity of membrane enzyme Mg2+-ATPase of the rabbit small intestinal brush border membrane. Their relationships with the changes in the fluidity of the membrane lipid bilayer were examined through studies on the fluorescence anisotropies of diphenylhexatriene (DPH) and its ionic derivatives. Good linear correlations were found both between the partition coefficients of the alcohols and their concentrations causing similar decreases in the activity of Mg2+-ATPase and between their partition coefficients and the alcohol-induced changes in fluorescence anisotropies. Within the concentration range of the alcohols tested, including isoamyl alcohol and benzyl alcohol, the decreases in activity of the membrane enzyme Mg2+-ATPase clearly corresponded with the decreases in fluorescence anisotropy of DPH, which is thought to be located within the hydrophobic core of the membrane. From these findings, one possible explanation is that inhibition of this enzyme by the alcohols is due to perturbation of the lipid bilayer of the brush border membrane.
Similar content being viewed by others
References
Chin, J.H., Goldstein, D.B. 1981. Membrane-disordering action of ethanol. Variation with membrane cholesterol content and depth of the spin label probe. Mol. Pharmacol. 19:425–431
Chin, J.H., Goldstein, D.B. 1985. Effects of alcohols on membrane fluidity and lipid composition. In: Membrane Fluidity in Biology Vol. 3. R.C. Aloia and J.M. Boggs, editors, pp. 1–38, Academic Press, New York
Devés, R., Krupka, R.M. 1990. Inhibition of choline transport in erythrocytes by n-alkanols. Biochim. Biophys. Acta 1030:32–40
Dickinson, R., Franks, N.P., Lieb, W.R. 1993. Thermodynamics of anesthetic/protein interactions. Temperature studies on firefly lucifarase. Biophys. J. 64:1264–1271
Fernandez, Y.J., Boigergrain, R.-A.M., Cambon-Gros, C.D., Mitjavila, S.E. 1984. Sensitivity of Na+-coupled d-glucose uptake, Mg2+-ATPase and sucrase to perturbations of the fluidity of brush-border membrane vesicles induced by n-aliphatic alcohols. Biochim. Biophys. Acta 770:171–177
Fiske, C., SubbaRow, J. 1926. The colorimetric determination of phosphorus. J. Biol. Chem. 66:375–400
Friedlander, G., Le Grimellec, C., Giocondi, M.-C., Amiel, C. 1987. Benzyl alcohol increases membrane fluidity and modulates cyclic AMP synthesis in intact renal epitherial cells. Biochim. Biophys. Acta 903:341–348
Garda, H.A., Brenner, R.R. 1984. Short-chain aliphatic alcohols increase rat-liver microsomal membrane fluidity and affect the activities of some microsomal membrane-bound enzymes. Biochim. Biophys. Acta 769:160–170
Gordon, L.M., Sauerheber, R.D., Esgate, J.A., Dipple, I., Marchmont, R.J., Houslay, M.D. 1980. The increase in bilayer fluidity of rat liver plasma membranes achieved by the local anesthetic benzyl alcohol affects the activity of intrinsic membrane enzymes. J. Biol. Chem. 255:4519–4527
Hansch, C., Dunn, III, W.J. 1972. Linear relationships between lipophilic character and biological activity of drugs. J. Pharm. Sci. 61:1–19
Hauser, H., Gains, N., Semenza, G., Spiess, M. 1982. Orientation and motion of spin-labels in rabbit small intestinal brush border vesicle membranes. Biochemistry 21:5621–5628
Jain, M.K., Gleeson, J., Upreti, A., Upreti, G.C. 1978. Intrinsic perturbing ability of alkanols in lipid bilayers. Biochim. Biophys. Acta 509:1–8
Kessler, M., Acuto, O. Storelli, C., Murer, H., Müller, M., Semenza, G. 1978. A modified procedure for the rapid preparation of efficiently transporting vesicles from small intestinal brush border membranes. Their use in investigating some properties of d-glucose and choline transport systems. Biochim. Biophys. Acta 506:136–154
Kitagawa, S., Hirata, H. 1992. Effects of alcohols on fluorescence anisotropies of diphenylhexatriene and its derivatives in bovine blood platelets: Relationships of the depth-dependent change in membrane fluidity by alcohols with their effects on platelet aggregation and adenylate cyclase activity. Biochim. Biophys. Acta 1112:14–18
Kitagawa, S., Kametani, F., Tsuchiya, K., Sakurai, H. 1990. ESR analysis with long-chain alkyl spin labels in bovine blood platelets. Relationship between the increase in membrane fluidity by alcohols and phenolic compounds and their inhibitory effects on aggregation. Biochim. Biophys. Acta 1027:123–129
Kitagawa, S., Matsubayashi, M., Kotani, K., Usui, K., Kametani, F. 1991. Asymmetry of membrane fluidity in the lipid bilayer of blood platelets: Fluorescence study with diphenylhexatriene and analogs. J. Membrane Biol. 119:221–227
Kitagawa, S., Shinohara, T., Kametani, F. 1984. Effects of alcohols on ADP-induced aggregation and membrane fluidity of gel-filtered bovine blood platelets. J. Membrane Biol. 79:97–102
Kubina, M., Lanza, F., Cazenave, J.-P., Laustriat, G., Kuhry, J.-G. 1987. Parallel investigation of exocytosis kinetics and membrane fluidity changes in human platelets with the fluorescent probe, trimethylammonio-diphenylhexatriene. Biochim. Biophys. Acta 901:138–146
Kuhry, J.-G., Fonteneau, P., Duportail, G., Maechling, C., Laustriat, G. 1983. TMA-DPH: A suitable fluorescence polarization probe for specific plasma membrane fluidity studies in intact living cells. Cell Biophys. 5:129–140
Kutchai, H., Cooper, R.A., Forster, R.E. 1980. Erythrocyte water permeability. The effects of anesthetic alcohols and alterations in the level of membrane cholesterol. Biochim. Biophys. Acta 600:542–552
Le Grimellec, C., Friedlander, G., El Yandouzi, E.L.H., Zlatkine, P., Giocondi, M.-C. 1992. Membrane fluidity and transport properties in epithelia. Kidney Int. 42:825–836
Lipka, G., Op den Kamp, J.A.F., Hauser, H. 1991. Lipid asymmetry in rabbit small intestinal brush border membrane as probed by an intrinsic phospholipid exchange protein. Biochemistry 30:11828–11836
Lopes, C.M.B., Louro, S.R.W. 1991. The effects of n-alkanols on the lipid/protein interface of Ca2+-ATPase of sarcoplasmic reticulum vesicles. Biochim. Biophys. Acta 1070:467–473
Mitjavila, S., Lacombe, C., Carrera, G. 1976. Changes in activity of rat brush border enzymes incubated with a homologous series of aliphatic alcohols. Biochem. Pharmacol. 25:625–630
Orme, F.W., Moronne, M.M., Macey, R.I. 1988. Modification of the erythrocyte membrane dielectric constant by alcohols. J. Membrane Biol. 104:57–68
Ponder, E., Hyman, C. 1939. Acceleration of hemolysis in relation to chemical structure II. The straight chain alcohols. Proc. Soc. Exp. Biol. Med. 42:320–322
Pringle, M.J., Brown, K.B., Miller, K.W. 1981. Can the lipid theories of anesthesia account for the cutoff in anesthetic potency in homologous series of alcohols. Mol. Pharmacol. 19:49–55
Schwichtenhövel, C., Deuticke, B., Haest, C.W.M. 1992. Alcohols produce reversible and irreversible acceleration of phospholipid flip-flop in the human erythrocyte membrane. Biochim. Biophys. Acta 1111:35–44
Stubbs, C.D., Smith, A.D. 1984. The modification of mammalian membrane polyunsaturated fatty acid composition in relation to membrane fluidity and function. Biochim. Biophys. Acta 779:89–137
Trotter, P.J., Storch, J. 1989. 3-[p-(6-Phenyl)-1,3,5-hexatrienyl]-phenylpropionic acid (PA-DPH): Characterization as a fluorescent membrane probe and binding to fatty acid binding proteins. Biochim. Biophys. Acta 982:131–139
Winpenny, J.P., Elliot, J.R., Harper, A.A. 1992. Effects of n-alkanols and a methyl ester on a transient potassium (IA) current in identified neurones from helix aspersa. J. Physiol. 456:1–17
Author information
Authors and Affiliations
Additional information
The authors thank M. Takano, PhD and Y. Tomita, PhD, Department of Pharmacy, University Hospital of Kyoto University, for instruction in preparation of the brush border membrane vesicles. This work was supported in part by grants from the Japanese Ministry of Education, Science and Culture (05671795 and 06304044) and Takeda Science Foundation.
Rights and permissions
About this article
Cite this article
Kitagawa, S., Sugaya, Y., Nishizawa, M. et al. Relationship of alcohol-induced changes in Mg2+-ATPase activity of rabbit intestinal brush border membrane with changes in fluidity of its lipid bilayer. J. Membarin Biol. 146, 193–199 (1995). https://doi.org/10.1007/BF00238008
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00238008