Abstract
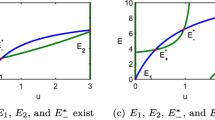
Although the dynamic behavior of microbial populations in nonmixed systems is a central aspect of many problems in biochemical engineering and microbiology, the factors that govern this behavior are not well understood. In particular, the effects of bacterial chemotaxis (biased migration of cells in the direction of chemical concentration gradients) have been the subject of much speculation but very little quantitative investigation. In this paper, we provide the first theoretical analysis of the effects of bacterial chemotaxis on the dynamics of competition between two microbial populations for a single rate-limiting nutrient in a confined nonmixed system. We use a simple unstructured model for cell growth and death, and the most soundly based current model for cell population migration. Using numerical finite element techniques, we examine both transient and steady-state behavior of the competing populations, focusing primarily on the influence of the cell random motility coefficient,μ, and the cell chemotaxis coefficient, χ. We find that, in general, there are four possible steady-state outcomes: both populations die out, population 1 exists alone, population 2 exists alone, and the two populations coexist. We find that, in contrast to well-mixed systems, the slower-growing population can coexist and even exist alone if it possesses sufficiently superior motility and chemotaxis properties. Our results allow estimation of the value of χ necessary to allow coexistence and predominance for reasonable values of growth and random motility parameters in common systems. An especially intriguing finding is that there is a minimum value of χ necessary for a chemotactic population to have a competitive advantage over an immotile population in a confined nonmixed system. Further, for typical system parameter values, this minimum value of χ is the range of values that can be estimated from independent experimental assays for chemotaxis.
Thus, in typical nonmixed systems, cell motility and chemotaxis properties can be the determining factors in governing population dynamics.
Similar content being viewed by others
References
Alt W (1980) Biased random walk models for chemotaxis and relted diffusion approximations. J Math Biol 9:147
Berg H (1983) Random walks in biology. Princeton University Press, Princeton, New Jersey
Boyce JR, Miller RV (1982) Motility as a selective force in the reversion of cystic fibrosis-associated mucoidPseudomonas aeruginosa to the nonmucoid phenotype in culture. Infect Immun 37:840
Boyd A, Simon M (1982) Bacterial chemotaxis. Ann Rev Physiol 44:501
Freter R, O'Brien PCM, Halstead SA (1978) Adhesion and chemotaxis as determinants of bacterial association with mucosal surfaces. Adv Exp Biol Med 107:4292
Hazelbauer GL, Harayama S (1983) Sensory transudction in bacterial chemotaxis. Int Rev Cytol 81:33
Holz M, Chen SH (1979) Spatio-temporal structure of migrating chemotactic bands ofEscherichia coli. Biophys J 26:243
Karel S, Libick S, Robertson CR (1985) The immobilization of whole cells: engineering principles. Chem Eng Sci 40:1321
Keller EF (1974) Mathematical aspects of bacterial chemotaxis. In: Sorkin E (ed) Chemotaxis: its biology and biochemistry. S. Karger, Basel, p 79
Keller EF, Segel LA (1971) Model for chemotaxis. J Theor Biol 30:225
Kelman A, Hruschka J (1973) The role of motility and aerotaxis in the selective increase of a virulent bacteria in still broth cultures ofPseudomonas solanacearum. J Gen Microbiol 76:177
Koshland DE Jr (1980) Bacterial chemotaxis as a model behavioral system. Raven Press, New York
Koshland DE Jr (1981) Biochemistry of sensing and adaptation in a simple bacterial system. Ann Rev Biochem 50:765
Lapidus IR, Schiller R (1978) A model for traveling bands of chemotactic bacteria. Biophys J 22:1
Lauffenburger DA, Aris R, Keller KH (1981) Effects of random motility on growth of bacterial populations. Microb Ecol 7:207
Lauffenburger DA, Aris R, Keller KH (1982) Effects of cell motility and chemotaxis on microbial population growth. Biophys J 40:209
Lauffenburger DA, Calcagno B (1983) Competition between microbial populations in a non-mixed environment: effect of cell random motility. Biotech Bioeng 25:2103
Lauffenburger DA, Kennedy CR, Aris R (1984) Traveling bands of chemotactic bacteria in the context of population growth. Bull Math Biol 46:19
Macnab RM (1978) Bacterial motility and chemotaxis: the molecular biology of a behavioral system. CRC Crit Rev Biochem 5:291
Mesibov R, Ordal GW, Adler J (1973) The range of attractant concentrations for bacterial chemotaxis and the threshold and size of reponse over this range. J Gen Physiol 62:203
Old DC, Duguid JP (1970) Selective outgrowth of fimbriate bacteria in static liquid medium. J Bacteriol 103:447
Pilgrim WR, Williams FD (1976) Survival value of chemotaxis in mixed cultures. Can J Microbiol 22:1771
Rivero M, Lauffenburger DA (1986) Analysis of the capillary assay for determination of Bacterial motility and chemotaxis parameters. Biotech Bioeng 28:1178
Rosen G (1978) Steady-state distribution of bacteria chemotactic toward oxygen. Bull Math Biol 40:671
Segel LA (1977) A theoretical study of receptor mechanisms in bacterial chemotaxis. SIAM J Appl Math 32:653
Segel LA, Jackson JL (1973) Theoretical analysis of chemotactic movement in bacteria. J Mechanochem Cell Motil 2:25
Segel LA, Chet I, Henis Y (1977) A simple quantitative assay for bacterial motility. J Gen Microbiol 98:329
Smith JL, Doetsch RN (1969) Studies on negative chemotaxis and the survial value of motility inPseudomonas fluorescens. J Gen Microbiol 55:379
Strang G, Fix GJ (1973) An analysis of the finite element method. Prentice-Hall, Englewood Cliffs, New Jersey
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kelly, F.X., Dapsis, K.J. & Lauffenburger, D.A. Effect of bacterial chemotaxis on dynamics of microbial competition. Microb Ecol 16, 115–131 (1988). https://doi.org/10.1007/BF02018908
Issue Date:
DOI: https://doi.org/10.1007/BF02018908