Summary
Determination of Calcium in Chlor-alkali Brine by Flow Injection Analysis
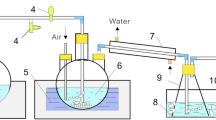
A few milligrams of calcium in one liter of chlor-alkali brine were determined by flow injection analysis. When the calcium content was lower, the preconcentration and separation of calcium from sodium chloride with a chelating resin column was employed. The flow injection analysis was based on the exchange reaction between calcium and zinc complex of ethylene glycol-bis(2-aminoethylether)tetraacetic acid (EGTA) in the presence of 2-(3,5-dibromo-2-pyridylazo)-5-[N-ethyl-N-(3-sulfopropyl)amino]phenol(H2R):
. Calcium (0.25–2.0 mg 1−1 in the presence of magnesium, iron(III), aluminum, copper(II), nickel, phosphate, chloride, sulfate and carbonate can be determined with a sampling rate of 40 h−1.
Similar content being viewed by others
References
G. Nakagawa, H. Wada, and C. Wei, Anal. Chim. Acta145, 135 (1983).
T. Uchida, C. Wei, C. Iida, and H. Wada, Nagoya Kogyo Daigaku Gakuho33, 97 (1982).
K. Ohshita, H. Wada, and G. Nakagawa, Anal. Chim. Acta149, 269 (1983).
H. Wada, T. Ishizuki, and G. Nakagawa, Mikrochim. Acta [Wien]1983 II, 235.
H. Wada, G. Nakagawa, and K. Ohshita, Anal. Chim. Acta159, 289 (1984).