Abstract
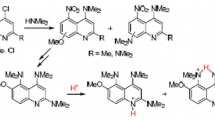
The pyrano-cyclisation of 4-hydroxy-(1-methyl)-1H-quinoline-2-one with 3-chlor-3-methylbut-1-yne, supposed to proceed via an ether or vinyl intermediate, is studied by alkylating different 4-hydroxyquinolines. The nine derivatives characterized favour the ether-pathway (“Späth type” chromene synthesis).
Similar content being viewed by others
Literatur
6. Mitt.: [7] zugleich Naturstoffchemie 113. Mitt.; 112. Mitt.:Reisch J. Podpetschnig E (1987) Pharmazie 42: 745
Teil der geplanten DissertationSalehi-Artimani RA (1988) Münster
Teil der DissertationMüller M (1981) Münster
Reisch J, Bathe A, Salehi-Artimani RA (1986) Arch Pharm (Weinheim) 319: 720 und dort zit. Literatur
Reisch J, Bathe A, Rosenthal BHW, Salehi-Artimani RA (1987) J Heterocycl Chem 24: 869
Reisch J, Bathe A (1987) Arch Pharm (Weinheim) 320: 737
Reisch J, Bathe A (1987) J Heterocycl Chem 24: 1409
Zimmermann HE, Mais A (1959) J Am Chem Soc 81: 3644
DissertationScheer M (1986) Münster
Späth E, Hittel R (1939) Chem Ber 72: 963 und 2093
Hansen HJ, Hesse M, Philipsborn W v (1978) Helv Chim Acta 61: 1
Reisch J, Dharmaratne HRW (1985) Z Naturforsch 40b: 636
Koller G (1927) Ber Dtsch Chem Ges 60: 1108
Reisch J, Müller M, Mester I (1981) Z Naturforsch 36b: 1176
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Reisch, J., Salehi-Artimani, R.A., Bathe, A. et al. Acetylenchemie 7. Mitt.: Mechanistische Studien zur Synthese von Pyrano-[3,2-c]chinolin-Alkaloiden aus 4-Hydroxychinolin-2-onen und 3-Chlor-3-methylbut-1-in. Monatsh Chem 119, 781–791 (1988). https://doi.org/10.1007/BF00809691
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00809691