Summary
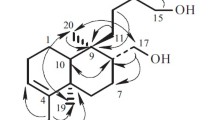
C15H24O2,M=236.2, monoclinic, P 21,a=7.229(7),b=14.925 (9),c=6.235 (9) Å, β=92.40 (9)°,V=672.1 Å3,T=−133 °C,Z=2,D x=1.17 g cm−3. The X-ray diffraction analysis of this sesquiterpenoid fromCarissa opaca confirmed the previously proposed constitution of the isolate and, furthermore, allowed precise NMR assignment.
Zusammenfassung
C15H24O2,M=236.2, monoklin, P 21,a=7.229 (7),b=14.925 (9),c=6.235 (9) Å, β=92.40 (9)°,V=672.1 Å3,T=−133 °C,Z=2,D x=1.17 g cm−3. Die Röntgenstrukturanalyse dieses Sesquiterpenoids ausCarissa opaca bestätigte die bereits vorgeschlagene Struktur und erlaubte ferner eine genaue NMR-Zuordnung.
Similar content being viewed by others
References
For Part 99 see: Adesina S. K., Reisch J. (1985) Phytochemistry24: 3003
Mohr K., Schindler O., Reichstein T. (1954) Helv. Chim. Acta37: 462
Achenbach H., Waibel R., Addae-Mensah I. (1983) Phytochemistry22: 749
Kutney J. P., Singh A. K. (1982) Can. J. Chem.60: 1842
Rao I. M., Swamy P. M., Das V. S. R. (1980) Z. Pflanzenphysiol.99: 69
Chopra R. N., Nayar S. L., Chopa I. C. (1956) Glossary of Indian Medicinal Plants. C.S.I.R., New Delhi, India, p. 52
Pakrashi S. C., Datta S., Ghosh-Dastidar P. P. (1968) Phytochemistry7: 495
Ward E. W. B., Unwin C. H., Hill J., Stoessl A. (1975) Phytopathol.65: 859
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Reisch, J., Hussain, R.A., Krebs, B. et al. Natural product chemistry, part 100: The structure of carissone, C15H24O2 . Monatsh Chem 121, 941–944 (1990). https://doi.org/10.1007/BF00808958
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00808958