Summary
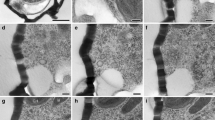
Immunofluorescence and TEM studies of meiosis in two mosses (Bryophyta) provide evidence that the prophasic tetrahedral system of microtubules contributes directly to the metaphase I spindle. Intense staining of tubulin, conspicuously absent around the nuclear envelope, is first seen associated with plastids. By mid-prophase, microtubules radiate from the plastids to the nuclear envelope and become organized into six bands that interconnect the four plastids, forming a tetrahedral cytoskeleton surrounding the nucleus. During transition of prophase to metaphase, the four poles of the tetrahedral microtubule system converge in pairs toward opposite cleavage furrows. Opposite furrows occupy mutually perpendicular planes and the pair of microtubule focal points straddling one furrow lies at right angles to the pair straddling the opposite furrow. Additional microtubules terminate in numerous small clusters in the concave polar regions arching over the cleavage furrows. By early anaphase, the microtubule focal points lie very close to the division axis. We conclude that microtubules recruited from the prophasic quadripolar system are incorporated into the mature metaphase I spindle and the two principal focal points at each pole are those derived from poles of the prophasic quadripolar system.
Similar content being viewed by others
References
Allen CE (1916) Four-lobed spore mother cells inCatharinea. Am J Bot 8: 456–460
Apostolakos P, Galatis B (1985) Studies on the development of the air pores and air chambers ofMarchantia paleacea III. Microtubule organization in preprophase-prophase initial aperture cells-formation of incomplete preprophase microtubule bands. Protoplasma 128: 120–135
Bajer A, Molè-Bajer J (1972) Spindle dynamics and chromosome movements. Int Rev Cytol [Suppl] 3. Academic Press, New York and London
— (1975) Lateral movements in the spindle and the mechanism of mitosis. In:Inoue S, Stephens RE (eds) Molecules and cell movement. Raven Press, New York, pp 77–96
— (1982) Asters, poles and transport properties within spindlelike microtubule arrays. Cold Spring Harbor Symp Quant Biol 46: 263–283
Brown RC, Lemmon BE (1982 a) Ultrastructure of meiosis in the mossRhynchostegium serrulatum I. Prophasic microtubules and spindle dynamics. Protoplasma 110: 23–33
— (1982 b) Ultrastructural aspects of moss meiosis: review of nuclear and cytoplasmic events during prophase. J Hattori Bot Lab 53: 29–39
— (1984) Plastid apportionment and preprophase microtubule bands in monoplastidic root meristem cells ofIsoetes andSelaginella. Protoplasma 123: 95–103
— (1985 a) Development of stomata inSelaginella: Division polarity and plastid movements. Am J Bot 72: 1914–1925
— (1985 b) Preprophasic establishment of division polarity in monoplastidic mitosis of hornworts. Protoplasma 124: 175–183
— (1985 c) A cytoskeletal system predicts division plane in meiosis ofSelaginella. Protoplasma 127: 101–109
Burgess J, Northcote DH (1968) The relationship between the endoplasmic reticulum and microtubular aggregation and disaggregation. Planta 80: 1–14
Clapham DH, Ostergren G (1984) Immunocytochemistry of tubulin at meiosis inTradescantia by a protein-A gold method. Hereditas 101: 137–142
Davis BM (1901) Nuclear studies onPellia. Ann Bot 15: 147–180
De Mey J, Lambert A-M, Bajer AS, Moeremans M, DeBrabander M (1982) Visualization of microtubules in interphase and mitotic plant cells ofHaemanthus endosperm with the immuno-gold staining method. Proc Natl Acad Sci USA 79: 1898–1902
Doonan JH, Cove DJ, Lloyd CW (1985) Immunofluorescence microscopy of microtubules in intact cell lineages in the moss,Physcomitrella patens I. Normal and CIPC-treated tip cells. J Cell Sci 75: 131–147
Eleftheriou EP (1985) Microtubules and root protophloem ontogeny in wheat. J Cell Sci 75: 165–179
Esau K, Gill RH (1969) Structural relations between nucleus and cytoplasm during mitosis inNicotiana tabacum mesophyll. Can J Bot 47: 581–591
Farmer JB (1894) Studies onHepaticae: onPallavicinia decipiens, Mitten. Ann Bot 8: 36–52 + 39 figures
— (1895) On spore-formation and nuclear division in theHepaticae. Ann Bot 9: 469–529 + 74 figures
— (1904) On the interpretation of the quadripolar spindle in theHepaticae. Bot Gaz 37: 63–65
Fowke LC, Pickett-Heaps JD (1978) Electron microscope study of vegetative cell division in two species ofMarchantia. Can J Bot 56: 467–475
Gunning BES (1982) The cytokinetic apparatus: Its development and spatial regulation. In:Lloyd CW (ed) The cytoskeleton in plant growth and development. Academic Press, London New York, pp 229–292
Hogan CJ (1985) Absence of pre-prophase band microtubules in meiotic higher plant cells. J Cell Biol 101 (5,2): 149 a
Kubiak J, de Brabander M, de Mey J, Tarkowska JA (1986) Origin of the mitotic spindle in onion root cells. Protoplasma 130: 51–56
Lambert A-M (1974) Ultrastructure de l'appareil fusorial en meiose chez la mousseMnium hornum L. Soc Bot Fr Coll Bryologie 121: 93–96
— (1977) La meiose chez lesBryophytes: ultrastructure et dynamique du fuseau chez une mousse,Mnium hornum Hedw. Bryophyt Biblio 13: 113–145
— (1980) The role of chromosomes in anaphase trigger and nuclear envelope activity in spindle formation. Chromosoma 76: 295–308
—,Bajer A (1972) Dynamics of spindle fibers and microtubules during anaphase and phragmoplast formation. Chromosoma 39: 101–144
— (1975) Fine structure dynamics of the prometaphase spindle. J Microsc Biol Cell 23: 181–194
Lander CA (1935) The relation of the plastid to nuclear division inAnthoceros laevis. Am J Bot 22: 42–51
Mitchison TJ, Kirschner MW (1984) Dynamic instability of microtubule growth. Nature 312: 237–241
— (1985) Properties of the kinetochorein vitro. II. Microtubule capture and ATP-dependent translocation. J Cell Biol 101: 766–777
Moore AC (1905) Sporogenesis inPallavicinia. Bot Gaz 40: 81–96
Pickett-Heaps JD (1969 a) Preprophase microtubules and stomatal differentiation; some effects of centrifugation on symmetrical and asymmetrical cell division. J Ultrastruct Res 27: 24–44
— (1969 b) Preprophase microtubules and stomatal differentiation inCommelina cyanea. Aust J Biol Sci 22: 375–391
— (1974) Plant microtubules. In:Robards AW (ed) Dynamic aspects of plant ultrastructure. McGraw-Hill, London New York, pp 219–255
Sakai A (1969) Electron microscopy of dividing cells. II. Microtubules and formation of the spindle in root tip cells of higher plants. Cytologia 34: 57–70
Schmit A-C, Vantard M, de Mey J, Lambert A-M (1983) Aster-like microtubule centers establish spindle polarity during interphase-mitosis transition in higher plant cells. Pl Cell Rep 2: 285–288
Schnepf E (1973) Mikrotubulus-Anordnung und -Umordnung, Wandbildung und Zellmorphogenese in jungenSphagnum-Blättchen. Protoplasma 78: 145–173
— (1984) Pre- and postmitotic reorientation of microtubule arrays in youngSphagnum leaflets: transitional stages and initiation sites. Protoplasma 120: 100–112
Singh AP (1977) The subcellular organization of stomatal initials in sugarcane leaves: the guard and subsidiary mother cells. Can J Bot 55: 2801–2809
Sorsa V (1955) Outlines of meiosis in the moss genusSphagnum. Hereditas 41: 250–258
Steer MW (1984) Mitosis inBryophytes. Advances in Bryology 2: 1–23 + 27 figures
Van Lammeren AAM, Keijzer CJ, Willemse MTM, Kieft H (1985) Structure and function of the microtubular cytoskeleton during pollen development inGasteria verrucosa (Mill.) H. Duval. Planta 165: 1–11
Wick SM, Duniec J (1983) Immunofluorescence microscopy of tubulin and microtubule arrays in plant cells. I. Preprophase band development and concomitant appearance of nuclear envelope-associated tubulin. J Cell Biol 97: 235–243
— (1984) Immunofluorescence microscopy of tubulin and microtubule arrays in plant cells. II. Transition between the preprophase band and the mitotic spindle. Protoplasma 122: 45–55
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brown, R.C., Lemmon, B.E. Division polarity, development and configuration of microtubule arrays in Bryophyte meiosis. Protoplasma 137, 84–99 (1987). https://doi.org/10.1007/BF01281144
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01281144