Summary
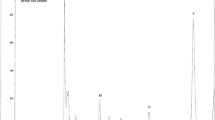
We have reported preparations and gas chromatographic analyses of volatile derivatives of sulfuric acid and taurine (Masuoka et al., 1988; 1989). By extending these studies, we have developed a method for the gas chromatographic determination of cysteic acid. Cysteic acid was converted to the N-isobutoxycarbonyl derivative by the reaction with isobutyl chloroformate in the presence of sodium hydroxide. After desalting with a cation-exchange column, the derivative was converted to the silver salt by reacting with silver oxide. The resulting silver salt was quantitatively esterified with methyl iodide in the presence of dimethyl sulfate and silver oxide. Dimethyl N-isobutoxy-carbonylcysteate [methyl 2-(N-isobutoxycarbonylamino)-3-(methoxysulfonyl) propanoate] formed was analyzed by gas chromatography. The calibration curve was linear up to 5.0µmol per ml of cysteic acid and the recovery was more than 95%.
Similar content being viewed by others
References
Kataoka H, Ohishi K, Makita M (1986) J Chromatogr 354: 482–485
Kinnier WJ, Wilson JE (1977) J Chromatogr 135: 508–510
Kuriyama K, Tanaka Y (1987) In: Jakoby, Griffith (eds) Methods in enzymology, vol 143. Academic Press, New York, pp 164–166
MacKenzie SL, Finlayson AJ (1980) J Chromatogr 187: 239–243
Masuoka N, Ubuka T, Akagi R, Yao K, Ishino K (1989) Acta Med Okayama 43: 253–259
Masuoka N, Ubuka T, Kinuta M, Yoshida S, Taguchi T (1988) Acta Med Okayama 42: 247–252
Shahrokhi F, Gehrke CW (1968) J Chromatogr 36: 31–41
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Masuoka, N., Ubuka, T., Yao, K. et al. Preparation and analysis of a volatile derivative of cysteic acid. Amino Acids 1, 375–378 (1991). https://doi.org/10.1007/BF00814006
Issue Date:
DOI: https://doi.org/10.1007/BF00814006