Abstract
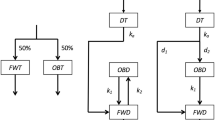
MORPHINE has been labelled with carbon-14 in the N-methyl position by chemical synthesis1,2 and randomly by biosynthesis3,4. Because of the metabolic lability of the N-methyl group and the need for considerable quantities of a product of high specific activity, studies on the preparation of tritium-nuclear labelled morphine were initiated. The Wilzbach procedure5 of exposure to tritium gas was utilized for the introduction of the tracer element into the morphine molecule. The preparation of biologically stable tritium-labelled morphine by this technique had been described previously6, and the radiochemical purity of the product was reported. We have found, however, that the exposure to tritium gas of morphine base or morphine sulphate in finely divided form, and subsequent purification to remove the labile tritium and radio-decomposition products, uniformly gave a seemingly pure crystalline morphine which showed two distinct areas of radioactivity when paper chromatograms were developed using correctly buffered paper and the proper solvent mixtures. One area contained a greater proportion of radioactivity (approximately 65–70 per cent of total) but negligible mass of material (not detectable by spray reagents or by visualization with ultra-violet light, less than 1 per cent). Similarly when normorphine base or its hydrochloride was tritiated in the same manner and purified, the morphine synthesized from it by the method of Andersen and Woods1 showed the same trace radio contaminant. When paper chromatograms were prepared from the above tritium-labelled ‘morphine’ using non-buffered paper and several solvent mixtures, that is, n-butanol, formic acid, and water (12 : 1 : 7 v/v); 2.3 per cent p-toluene sulphonic acid in n-butanol and water (100: 14-v/v), etc., all the radioactivity was located in one area. This area was the alkaloidal spot as localized by spraying with iodoplatinate alkaloidal reagent or by visualization of the shadow with ultra-violet light. The R F values were identical with parallel non-labelled morphine.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Andersen, K. S., and Woods, L. A., J. Org. Chem., 24, 274 (1959).
Rapoport, H., Lovell, C. H., and Tolbert, B. M., J. Amer. Chem. Soc., 73, 5900 (1951).
Battersby, A. R., and Harper, B. J. T., Chem. and Ind., 363 (1958). Leete, E., ibid., 977 (1958).
Achor, L. B., and Geiling, E. M. K., Anal. Chem., 26, 1061 (1954).
Wilzbach, K. E., J. Amer. Chem. Soc., 79, 1013 (1957).
Achor, L. B., J. Pharmacol. Exp. Therap., 122, 1A (1958).
Brossi, A., Häflinger, O., and Schnider, O., Arzneim. Forsch., 5, 62 (1955).
Reichstein, T., and Shoppee, C. W., Discuss. Farad. Soc., 7, 305 (1949).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
MISRA, A., WOODS, L. Preparation of Radiochemically Pure Tritium Nuclear-labelled Morphine. Nature 185, 304–305 (1960). https://doi.org/10.1038/185304b0
Issue Date:
DOI: https://doi.org/10.1038/185304b0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.