Abstract
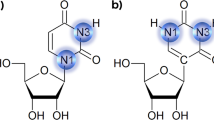
THE allowed conformations of nucleic acids depend on the flexibility of the structure of the nucleotides from which they are constructed. Many X-ray crystallographic studies of the degree of flexibility of these structures have shown that nucleosides exhibit a larger number of preferred conformations than nucleotides, and this led Sundaralingam1 to suggest that a nucleotide is more ‘rigid’ than a nucleoside. Berthod and Pullman2, however, have drawn attention to the fact that considerations of conformational energy do not imply such a rigid structure.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Sundaralingam, M., The Jerusalem Symposium on Quantum Chemistry and Biochemistry, 5 (edited by Bergmann, E. D., and Pullman, B.) (The Israel Academy of Sciences and Humanities, Jerusalem, 1972).
Berthod, H., and Pullman, B., FEES Lett., 30, 231 (1973).
Konnert, J., Karle, I. L., and Karle, J., Ada Cryst., B 26, 770 (1970).
Saenger, W., and Suck, D., Nature, 242, 610 (1973).
Chandrasekhar, K., McPherson, A., Adams, M. J., and Rossmann, M. G., J. molec. Biol, 76, 503 (1973).
Arnone, A., Bier, C. J., Cotton, F. A., Day, V. W., Hazen, E. E., Richardson, D. C., Richardson, J. S., and Yonath, A., J. biol. Chem., 246, 2302 (1971).
Richards, F. M., and Wyckoff, H. W., The Enzymes, IV (edit. by Boyer, P. D.). (Academic Press, 1971).
Crick, F. H. C., and Watson, J. D., Proc. R. Soc., A 223, 80 (1954).
Langridge, R., Marvin, D. A., Seeds, W. E., Wilson, H. R., Hooper, C. W., Wilkins, M. H. F., and Hamilton, L. D., J. molec. Biol., 2, 38 (1960).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
YOUNG, D., TOLLIN, P. & WILSON, H. Molecular conformation of deoxyguanosine 5′-phosphate. Nature 248, 513–514 (1974). https://doi.org/10.1038/248513a0
Received:
Issue Date:
DOI: https://doi.org/10.1038/248513a0
This article is cited by
-
Is 3′-nucleotide rigid?
Nature (1980)
-
Nucleotide rigidity
Nature (1976)
-
Structure of thymidylyl-3′, 5′-deoxyadenosine
Nature (1976)
-
Molecular structures of cytidine-5′-diphosphate and cytidine-5′-diphospho-choline, and their role in intermediary metabolism
Nature (1975)
-
An uncommon nucleotide conformation shown by molecular structure of deoxyuridine-5′-phosphate and nucleic acid stereochemistry
Nature (1975)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.