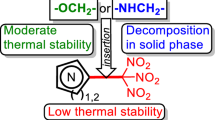
Summary
-
1.
The thermal stability of heterocyclic sulfur compounds has been studied by means of derivatography and isothermal gravimetry. It has been shown that the temperature for initial rupture of the five-membered ring decreases upon going from 3-phenyl-5-ethyl-4-thioxo-2-thiazolidone to 3-phenyl-4-thioxo-2-thiazolidone. The weight losses upon heating (isothermal conditions) increase in this same order.
-
2.
It has been established that the addition of metals catalyzes the breakdown process for 4-thioxo-2-thiazolidones.
-
3.
It has been shown that splitting of the thiazolidone ring takes place at the sulfur-containing junction, with the formation of dimerized nitrogen-containing compounds.
Similar content being viewed by others
Literature cited
US Pat. 2,711,393 (1955).
US Pat. 2,828,260 (1958).
US Pat. 2,971,911 (1961).
US Pat. 2,907,646 (1959).
V. I. Isagulyants and R. S. Boeva, Proceedings of Second All-Union Scientific-Technical Conference [in Russian], Khimiya, Moscow (1968).
H. Allcock, Heteroatom Ring Systems and Polymers, Academic Press, New York (1967).
L. J. Bellamy, Advances in Infrared Group Frequencies, Halsted, New York (1968).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya i Tekhnologiya Topliv i Masel, No. 8, pp. 15–18, August, 1974.
Rights and permissions
About this article
Cite this article
Sarnavskaya, T.I., Gordash, Y.T. Thermal stability of heterocyclic sulfur compounds. Chem Technol Fuels Oils 10, 601–604 (1974). https://doi.org/10.1007/BF00726369
Issue Date:
DOI: https://doi.org/10.1007/BF00726369