Abstract
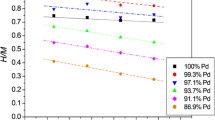
Amorphous ternary palladium-based alloys containing platinum group metals as an additional element were prepared by rapid quenching from the molten state and their anodic characteristics were investigated in a 4 mol dm−3 NaCl solution of pH 4 and 80° C. The amorphous alloys containing sufficient quantities of rhodium, platinum or iridium were passivated by anodic polarization and their corrosion rates at high current densities in the chlorine evolution region were extremely low. This fact was attributed to the formation of a highly protective passive film due to both the transformation to the amorphous structure and the addition of rhodium, platinum or indium. The electrocatalytic activities for chlorine evolution of amorphous alloys were higher than those of pure platinum group metals except palladium. In particular, the amorphous Pd41Ir40P19 alloy had the desired stable, high electrocatalytic activity for chlorine evolution and the high overvoltage for oxygen evolution.
Similar content being viewed by others
References
A. Yokoyama, H. Komiyama, H. Inoue, T. Masumoto and H. M. Kimura,Scripta Met. 15 (1981) 365.
Idem, J. Catalysis 68 (1981) 355.
Idem, Rapidly Quenched Metals in ‘Proceedings of the 4th Interntional Conference on Rapidly Quenched Metals’, Vol. II, (edited by T. Masumoto and K. Suzuki) Japan Institute of Metals, Sendai (1982) p. 1419.
A. Kawashima and K. Hashimoto,ibid. Japan Institute of Metals, Sendai (1982) p. 1427.
A. T. Kühn, ‘Industrial Electrochemical Processes’, Elsevier, Amsterdam (1971).
O. De Nora,Chem. Ind. Techn. 42 (1970) 222.
B. V. Ershier,Z. Fiz. Khim. 18 (1944) 131.
G. A. Tedoradse,Z. Fiz. Khim. 33 (1959) 129.
J. S. Mayell and S. H. Langer,Electrochim. Acta 9 (1964) 1411.
J. Llopis, J. M. Gamboa and J. M. Alfayate,Electrochim. Acta 12 (1967) 57.
‘Soda Handbook’, Japan Soda Industry Association, Tokyo, (1975) p. 224.
M. Takahashi,Soda and Chlorine 26 (1975) 267.
Ya. M. Kolotyrkin,Denki Kagaku 47 (1979) 390.
General Electric Co. Ltd. Japanese Patent 54-97581, 54-112398(1979).
M. Naka, K. Hashimoto and T. Masumoto,Corrosion 32 (1976) 146.
K. Hashimoto, M. Kasaya, K. Asami and T. Masumoto,Corros. Eng. (Boshoku Gijutsu) 26 (1977) 445.
M. Naka, K. Hashimoto and T. Masumoto,J. Non-Cryst. Solids 29 (1978) 61.
Idem, ibid. 31 (1979) 257.
Idem, ibid. 34 (1979) 347.
M. Naka, K. Hashimoto, A. Inoue and T. Masumoto,ibid. 31 (1979) 347.
T. Masumoto and R. Maddin,Acta Met. 19 (1971) 725.
M. Hara, K. Hashimoto and T. Masumoto,Electrochim. Acta 25 (1980) 1215.
M. Hara, K. Asami, K. Hashimoto and T. Masumoto,Electrochim. Acta 25 (1980) 1091.
D. Galizzioli, F. Tantardini and S. Trasatti,J. Appl. Electrochem. 4 (1974) 57.
M. Naka, K. Hashimoto and T. Masumoto,J. Non-Cryst. Solids 28 (1978) 403.
K. Hashimoto, M. Naka, K. Asami and T. Masumoto,Corros. Eng. (Boshoku Gijutsu) 27 (1978) 279.
A. A. Jakowkin,Z. physik. Chem. 29 (1899) 61.
M. Takahashi,Soda and Chlorine 29 (1978) 379.
K. Hashimoto, K. Osada, T. Masumoto and S. Shimodaira,Corros. Sci. 16 (1976) 71.
K. Hashimoto, K. Asami, M. Naka and T. Masumoto,Corros. Eng. (Boshoku Gijutsu) 28 (1979) 271.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hara, M., Hashimoto, K. & Masumoto, T. Anodic characteristics of amorphous ternary palladium-phosphorus alloys containing ruthenium, rhodium, iridium, or platinum in a hot concentrated sodium chloride solution. J Appl Electrochem 13, 295–306 (1983). https://doi.org/10.1007/BF00941601
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00941601