Abstract
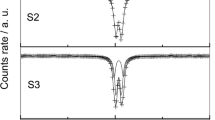
The crystallographic structure of Ni(II)−Fe(III) hydroxy-chloride green rusts obtained by oxidation of a FexNi1−x(OH)2 precipitate is isomorphous with that of the ferrousferric green rust one and independent of the value of P=Fe/Ni. Mössbauer spectra exhibit two quadrupole doublets after further oxidation of the compounds which correspond to a formula (3-x)Ni(OH)2 · xFeOOH · Fe(OH)2Cl. The Fe3+ ions are found to occupy preferentially the sites close to the Cl− ions and the Ni2+ those far from them. However the ordering of the Fe3+ ions is not perfect.
Similar content being viewed by others
References
J.D. Bernal, D.R. Dasgupta and A.L. Mackay, Clay Min. Bull. 4 21 (1959) 15.
J.M.R. Génin, D. Rézel, Ph. Bauer, A.A. Olowe and A. Béral, Electrochem. Methods in Corr. Research, Materials Science Forum 8 (1986) 477.
Ph. Refait, D. Rézel and J.M.R. Génin, this issue
“Applied X-rays”, George Clark, 4th edition (1955) Mc Graw Hill, New York.
“Physical Chemistry,” E.A. Moelwyn-Hughes, (1961) Pergamon Press, Oxford.
“Crystal Physics”, G. S. Zhdanov (1965) Oliver and Boyd.
“Handbook of Chemistry and Physics”, 68th edit. (1987–88) R.C. West editor, CRC Press.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Refait, P., Génin, J.M.R. The structure of Ni(II)−Fe(III) hydroxy-chloride green rusts by Mössbauer spectroscopy and X-ray diffraction. Hyperfine Interact 69, 827–830 (1992). https://doi.org/10.1007/BF02401954
Issue Date:
DOI: https://doi.org/10.1007/BF02401954