Abstract
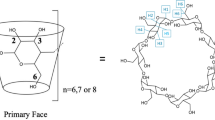
The conformational and binding properties of mono-6-deoxy-6[4-(1-ethyl-4-pyridinio)-1-pyridinio]-β-cyclodextrin (1) in complex formation with some guest compounds were examined by induced circular dichroism (ICD) spectra in aqueous solution. Compound 1 showed much stronger binding ability for some guest compounds (1: 1 complexes), compared withβ-cyclodextrin (β-CDx) and a positively charged β-CDx [C-6-mono-pyridino-β-CDx (2)]. Marked conformational changes of1 (the spatial position of the ethyl viologen (C2V2+) group relative to the cavity in 1) were observed upon complex formation with some guests like 1-adamantanecarboxylic acid (ACA) and sodium cholate (SC).
Similar content being viewed by others
References
M. L. Bender and M. Komiyama:Cyclodextrin Chemistry, Springer-Verlag (1978).
Y. Du, A. Nakamura and F. Toda:Bull. Chem. Soc. Jpn. 63, 3351 (1990).
M. Gratzel:Ber. Bunsenges. Phys. Chem. 84, 981 (1980).
I. Willner, D. Mandler and A. Riklin:J. Chem. Soc., Chem. Commun. 1986, 1022.
K. Takahashi, K. Hattori and F. Toda:Tetrahedron Lett. 25, 3331 (1984).
Y. Matsui, K. Ogawa, S. Mikami, M. Yoshimoto and K. Mochida:Bull. Chem. Soc. Jpn. 60, 1219 (1987); Y. Matsui and A. Okimoto:Bull. Chem. Soc. Jpn. 51, 3030 (1978).
K. Harata and H. Uedaira:Bull. Chem. Soc. Jpn. 48, 375 (1975); T. Murakami, K. Harata and S. Morimoto:Chem. Lett. 1989, 341; M. Kajtar, Cs. Horvath-Toro, E. Kuthi and J. Szejtli:Acta Chim. Acad. Sci. Hung. 110, 327 (1982).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Du, YQ., Nakamura, A. & Toda, F. The conformational and binding properties of ethylbipyridinio-modifiedβ-Cyclodextrin using induced circular dichroism. J Incl Phenom Macrocycl Chem 10, 443–451 (1991). https://doi.org/10.1007/BF01061075
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01061075