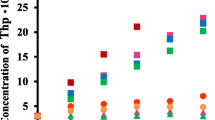
Abstract
Periodate oxidation followed by borohydride reduction of beta cyclodextrin results in crown ether type derivatives. The polyaldehyde obtained by periodate oxidation has been reduced to polyalcohol and converted to a permethylated derivative. These macrorings are much more flexible than beta cyclodextrin itself.
The interaction of beta cyclodextrin (β-CD), methylated beta cyclodextrin (DIMEB), polyalcohol and permethylated crown ethers derived from beta cyclodextrin with two triphenylmethane, derivatives and their triphenyltin analogues has been studied. On the basis of the changes in the UV spectra and the solubility enhancement it can be concluded that cyclodextrins (β-CD and DIMEB) form complexes only with the triphenylmethane, derivatives. On the other hand the crown ethers prepared from β-CD give complexes only with the triphenyltin derivatives.
Similar content being viewed by others
References
G. W. Hay, B. A. Lewis, and F. Smith:Methods Carb. Chem. 5, 377 (1965).
L. Kandra, Lipták, A., I. Jodál, P. Nánási, and J. Szejtli:J. Incl. Phenom. 2, 869 (1984).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Szejtli, J., Kandra, L. Crown ethers derived from cyclodextrin: Interaction with triphenylmethane derivatives. Journal of Inclusion Phenomena 5, 639–643 (1987). https://doi.org/10.1007/BF00663005
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00663005