Abstract
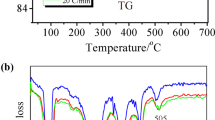
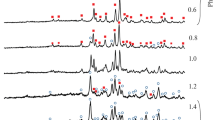
Zinc diphosphates (Zn2P2O7 · 5H2O, Zn2P2O7 · 3H2O, 5K1.4Zn1.3P2O7 · 16H2O, and 5K0.8Zn1.6P2O7 · 9H2O) were made by the wet process. The composition of the products was dependent on the conditions (concentration, pH, and dropping rate of the solution) of the process. When the products were heated, decomposition of the diphosphates to orthophosphate took place below about 150 ·C. Polymerization of the phosphates to phosphates with longer chains was observed in the temperature range 150 to 400 ° C. The amorphous phosphates thermally produced by heating diphosphates other than 5K1.4Zn1.3P2O7 · 16H2O, showed reorganization to diphosphate above 400 ° C according to the reaction M2O3PO[P(O2M)O] n PO3M2 +nM3PO4→(n + 1)M4P2O7 where M represents K and/or 1/2Zn.
Similar content being viewed by others
References
J. R. Van Wazer, “Phosphorus and Its Compounds”, Vol. II (Interscience, New York, 1961) p. 987.
B. A. Purin, “Electrodeposition of Metal in Electroplating Bath of Pyrophosphates” (Zinatne, Riga, 1975).
M. Watanabe, K. Tanabe, T. Takahara andT. Yamada,Bull. Chem. Soc. Jpn 44 (1971) 712.
Y. Hirai, N. Yoza andS. Ohashi,Anal. Chim. Acta 115 (1980) 269.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Watanabe, M., Onoda, S. The synthesis and thermal behaviour of zinc diphosphates. J Mater Sci 25, 4356–4360 (1990). https://doi.org/10.1007/BF00581095
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00581095