Abstract
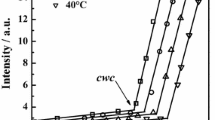
A narrow, reversible endothermic main transition is found in the aqueous micellar phase of octaethylene glycol tetradecyl ether (C14E8) by DSC, characterized by a transition temperature of 41°C and a ΔH value of 0.5 kcal mol−1, which is not observed by light scattering. This transition is assigned to a cooperative conformational rearrangement of the assembled amphiphilic detergent molecules and not to a micelle aggregation process. It is suggested that the detergent’s polar head group is primarily involved in this rearrangement.
Similar content being viewed by others
References
D. J. Mitchell, G. J. T. Tiddy, L. Waring, T. Bostock and M. P. McDonald, J. Chem. Soc., Farad. Trans. 1, 79 (1981) 975.
M. Zulauf, In Crystallization of Membrane Proteins (H. Michel ed.), CRC Press Boca Raton 1991, p. 53.
M. Kahlweit, Tenside Surf. Det., 30 (1993) 83.
H. Michel, In Crystallization of Membrane Proteins (H. Michel ed.), CRC Press, 1991, p. 73.
R. Strey, Ber. Bunsenges. Phys. Chem., 100 (1996) 182.
A. Blume, In Physical Properties of Membranes, Functional Implications (C. Hidalgo ed.), Plenum Press, New York 1988.
Manuscript in preparation.
Rights and permissions
About this article
Cite this article
Grell, E., Lewitzki, E., von Raumer, M. et al. Lipid-similar Thermal Transition of Polyethylene Glycol Alkyl Ether Detergents. Journal of Thermal Analysis and Calorimetry 57, 371–375 (1999). https://doi.org/10.1023/A:1010158312153
Issue Date:
DOI: https://doi.org/10.1023/A:1010158312153