Abstract
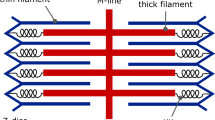
Despite extensive knowledge of many muscle A-band proteins (myosin molecules, titin, C-protein (MyBP-C)), details of the organization of these molecules to form myosin filaments remain unclear. Recently the myosin head (crossbridge) configuration in a relaxed vertebrate muscle was determined from low-angle X-ray diffraction (Hudson et al. (1997), J Mol Biol 273: 440–455). This showed that, even without C-protein, the myosin head array displays a characteristic polar pattern with every third 143 Å-spaced crossbridge level particularly prominent. However, X-ray diffraction cannot determine the polarity of the crossbridge array relative to the neighbouring actin filaments; information crucial to a proper understanding of the contractile event. Here, electron micrographs of negatively-stained goldfish A-segments and of fast-frozen, freeze-fractured plaice A-bands have been used to determine the resting myosin head polarity relative to the M-band. In agreement with the X-ray data, the prominent 429 Å-spaced striations are seen outside the C-zone, where no non-myosin proteins apart from titin are thought to be located. The head orientation is with the concave side of the curved myosin heads (containing the entrance to the ATP-binding site) facing towards the M-band and the convex surface (containing the actin-binding region at one end) facing away from the M-band.
Similar content being viewed by others
References
Al-Khayat HA, Yagi N and Squire JM (1995) Structural changes in actin-tropomyosin during muscle regulation: computer modelling of low-angle X-ray diffraction data. J Mol Biol 252: 611–632.
Bennett P, Craig R, Starr R and Offer G (1986) The ultrastructural location of C-protein, X-protein and H-protein in rabbit muscle. J Muscle Res Cell Motil 7: 550–567.
Chayen N, Freundlich A and Squire JM (1987) Comparative histo-chemistry of a flatfish fin muscle and of other vertebrate muscle used for ultrastructural studies. J Muscle Res Cell Motil 8: 358–371.
Chew MWK and Squire JM (1995) Packing of a-helical coiled-coil myosin rods in vertebrate muscle thick filaments. J Struct Biol 115: 233–249.
Cobb JLS, Fox NC and Santer RM (1973) A specific ringer solution for the plaice. J Fish Biol 5: 587–591.
Craig R (1977) Structure of A-segments from frog and rabbit skeletal muscle. J Mol Biol 109: 69–81.
Craig R and Offer G (1976) The location of C-protein in rabbit skeletal muscle. Proceedings of the Royal Society of London, Series B: Biological Sciences 192: 451–461.
Gilbert R, Cohen JA, Pardo S, Basu A and Fischman DA (1999) Identification of the A-band localization domain of myosin binding proteins C and H (MyBP-C, MyBP-H) in skeletal muscle. J Cell Sci 112: 69–79.
Hanson E, O'Brien EJ and Bennett PM (1971) Structure of the myosin-containing filament assembly (A-segment) separated from frog skeletal muscle. J Mol Biol 58: 865–871.
Harford J and Squire J (1986) ‘Crystalline’ myosin crossbridge array in relaxed bony fish muscle. Biophys J 50: 145–155.
Harford JJ and Squire JM (1992) Evidence for structurally different attached states of myosin crossbridges on actin during contraction of fish muscle. Biophys J 63: 387–396.
Herzberg O and James MNG (1985) Structure of the calcium regulatory muscle protein troponin-C at 2.8 Å resolution. Nature 313: 653–659.
Holmes KC, Popp D, Gebhard W and Kabsch W (1990) Atomic model of the actin filament. Nature 347: 44–49.
Hudson L, Harford JJ, Denny RC and Squire JM (1997) Myosin head configuration in relaxed fish muscle: resting state myosin heads must swing axially by up to 150 Å or Turn upside down to reach rigor. J Mol Biol 273: 440–455.
Huxley HE and Brown W (1967) The low-angle X-ray diagram of vertebrate striated muscle and its behavior during contraction and rigor. J Mol Biol 30: 383–434.
Improta S, Kruger JK, Gautel M, Atkinson RA, Lefevre JF, Moulton S, Trewhella J and Pastore A (1998) The assembly of immunoglubulin-like modules in titin: implications for muscle elasticity. J Mol Biol 284: 761–777.
Kabsch W, Mannherz HG, Suck D, Pai EF and Holmes KC (1990) Atomic structure of the actin: DNAase I complex. Nature 347: 37–44.
Kensler RW and Stewart M (1989) An ultrastructural study of crossbridge arrangement in the fish skeletal muscle thick filament. J Cell Science 94: 391–401.
Labeit S and Kolmerer B (1995) Titins: giant proteins in charge of muscle ultrastructure and elasticity. Science 270: 293–296.
Lehman W, Vilbert P, Uman P and Craig R (1995) Steric blocking by tropomyosin visualized in relaxed vertebrate thin filaments. J Mol Biol 251: 191–196.
Lorenz M, Poole K, Popp D, Rosenbaum G and Holmes K (1995) An atomic model of the unregulated thin filament obtained by X-ray fiber diffraction on oriented actin-tropomyosin gels. J Mol Biol 246: 108–119.
Luther PK, Munro PMG and Squire JM (1981) Three-dimensional structure of the vertebrate muscle A-band: 3. M-region structure and myosin filament symmetry. J Mol Biol 151: 703–730.
Luther PK, Munro PMG and Squire JM (1996a) Muscle ultrastructure in the teleost fish. Micron 26: 431–459.
Luther PK and Squire JM (1980) Three-dimensional structure of the vertebrate muscle A-band: 2. The myosin filament superlattice. J Mol Biol 141: 409–439.
Luther PK, Squire JM and Forey PL (1996b) Evolution of simple lattice and superlattice A-bands in vertebrate skeletal muscle. J Morph 229: 329–335.
Maruyama K (1997) Connectin/titin, giant elastic protein of muscle. FASEB Journal 11: 341–345.
Morris EP, Katayama E and Squire JM (1994) Evaluation of high-resolution shadowing applied to freeze-fractured, deep-etched particles: 3-D helical reconstruction of shadowed actin filaments. J Struct Biol 113: 47–55.
Rayment I, Rypniewski WR, Schmidt-Base K, Smith R, Tomchick DR, Benning MM, Winkelmann DA, Wesenberg G and Holden HM (1993) Three-dimensional structure of myosin subfragment-1: a molecular motor. Science 261: 50–58.
Rome E, Offer G and Pepe FA (1973) X-ray diffraction of muscle labelled with antibody to C-protein. Nature New Biol 244: 152–154.
Sjostrom M and Squire JM (1977a) Cryo-ultramicrotomy and myofibrillar fine structure: a review. J Micros 111: 239–278.
Sjostrom M and Squire JM (1977b) Fine structure of the A-band in cryo-sections. The structure of the A-band of human skeletal muscle fibers from ultrathin cryo-sections negatively stained. JMol Biol 109: 49–68.
Squire JM (1972) General model of the myosin filament structure II: myosin filaments and crossbridge interactions in vertebrate striated and insect flight muscles. J Mol Biol 72: 125–138.
Squire JM, Cantino M, Chew MWK, Denny RC, Harford JJ, Hudson L and Luther PK (1998) Myosin rod packing schemes in vertebrate muscle thick filaments. J Struct Biol 122: 128–138.
Squire JM, Harford JJ, Edman AC and Sjostrom M (1982) Fine structure of the A-band in cryo-sections III. Crossbridge distribution and the axial structure of the human C-zone. J Mol Biol 155: 467–494.
Trinick J (1996) Cytoskeleton: titin as a scaffold and spring. Current Biology 6: 258–260.
Vassylyev DG, Takeda S, Wakatsuki S, Maeda K and Maeda Y (1998) Crystal structure of troponin C in complex with troponin I fragment at 2.3 Å resolution. Proc Natl Acad Sci USA 95: 4847–4852.
Vibert P, Craig R and Lehman W (1997) Steric-model for activation of muscle thin filaments. J Mol Biol 266: 8–14.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cantino, M.E., Brown, L.D., Chew, M. et al. A-band architecture in vertebrate skeletal muscle: polarity of the myosin head array. J Muscle Res Cell Motil 21, 681–690 (2000). https://doi.org/10.1023/A:1005661123914
Issue Date:
DOI: https://doi.org/10.1023/A:1005661123914