Abstract
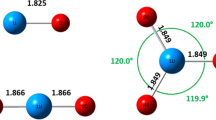
A comparative study of NH +4 formation on aluminosilicate surface and in an aqueous solution of hydrochloric acid has been carried out in the framework of the nonempirical method with a STO-3G basis. It is suggested that the compensation mechanisms of energy consumptions to form protonated species in both cases are similar.
Abstract
В рамках неэмпирического метода в базисе СТО-3Г про-ведено сравнительное исследование реакций образования NH +4 на поверхности алюмосиликата и в водном ра-створе соляной кислоты. Делается вывод о сходстве механизмов компенсации энергетических эатрат на об-разование протонированной формы на поверхности и в кислой среде.
Similar content being viewed by others
References
V.B. Kazanskii: Proc. 1st All-Union Conference Catalysis and Catalytic Processes in Production of Chemico-Pharmaceutical Compounds (Abstracts), p. 3 Moskva, All-Union Chemico-Technological Research Institute of Medical Industry, 1985.
A.G. Pelmenshchikov, V.I. Pavlov, G.M. Zhidomirov, Phys. Status Solidi B,130, 1 (1985).
E.A. Paukshtis, E.N. Yurchenko: Usp. Khim.,52, 426 (1983).
G.V. Tsitsishvili, D.V. Khuroshvili, G.M. Zhidomirov, A.G. Pelmenshchikov: React. Kinet. Catal. Lett.,25, 83 (1984).
L. Little: Infrared Spectra of Adsorbed Molecules. Mir, Moskva 1969.
E. Clementi: J. Chem. Phys.,46, 385;47, 2323 (1967).
Yu.D. Pankratiev, E.A. Paukshtis, V.M. Turkov, E.N. Yurchenko: Proc. Intern. Symposium on Zeolite Catalysis, p. 55, Siófok, Hungary 1985.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pelmenshchikov, A.G., Paukshtis, E.A., Zhanpeisov, N.U. et al. Comparative discussion of NH +4 formation mechanisms on oxide surfaces and in acidic media. React Kinet Catal Lett 33, 423–428 (1987). https://doi.org/10.1007/BF02128100
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02128100