Abstract
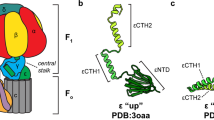
The b subunit of ATP synthase is a major component of the second stalk connecting the F1and F0 sectors of the enzyme and is essential for normal assembly and function. The156-residue b subunit of the Escherichia coli ATP synthase has been investigated extensivelythrough mutagenesis, deletion analysis, and biophysical characterization. The two copies ofb exist as a highly extended, helical dimer extending from the membrane to near the top ofF1, where they interact with the δ subunit. The sequence has been divided into four domains:the N-terminal membrane-spanning domain, the tether domain, the dimerization domain, andthe C-terminal δ-binding domain. The dimerization domain, contained within residues 60–122,has many properties of a coiled-coil, while the δ-binding domain is more globular. Sites ofcrosslinking between b and the a, α, β, and δ subunits of ATP synthase have been identified,and the functional significance of these interactions is under investigation. The b dimer mayserve as an elastic element during rotational catalysis in the enzyme, but also directly influencesthe catalytic sites, suggesting a more active role in coupling.
Similar content being viewed by others
REFERENCES
Abrahams, J. P., Leslie, A. G. W., Lutter, R., and Walker, J. E. (1994). Nature (London) 370, 621–628.
Angov, E., Ng, T. C. N., and Brusilow, W. S. A. (1991). J. Bacteriol. 173, 407–411.
Blair, A., Ngo, L., Park, J., Paulsen, I. T., and Saier, M. H. (1996). Microbiology 142, 17–32.
Boyer, P. D. (1997). Annu. Rev. Biochem. 66, 717–749.
Caviston, T. L., Ketchum, C. J., Sorgen, P. L., Nakamoto, R. K., and Cain, B. D. (1998). FEBS Lett. 429, 201–206.
Cherepanov, D. A., Mulkidhanian, A. Y., and Junge, W. (1999). FEBS Lett. 449, 1–6.
Cipriano, D. J. and Dunn, S. D. (2000) unpublished observations.
Collinson, I. R., van Raaij, M. J., Runswick, M. J., Fearnley, I. M., Skehel, J. M., Orriss, G. L., Miroux, B., and Walker, J. E. (1994). J. Mol. Biol. 242, 408–421.
Dmitriev, O., Jones, P. C., Jiang, W., and Fillingame, R. H. (1999). J. Biol. Chem. 274, 15598–15604.
Dunn, S. D., unpublished observations.
Dunn, S. D. (1992). J. Biol. Chem. 267, 7630–7636.
Dunn, S. D. and Chandler, J. (1998). J. Biol. Chem. 273, 8646–8651.
Dunn, S. D., Bi, Y., and Revington, M. (2000a). Biochim. Biophys. Acta, 1459, 521–527.
Dunn, S. D., Kellner, E., and Lill, H., manuscript submitted.
Dunn, S. D., McLachlin, D. T., and Revington, M. (2000b). Biochim. Biophys. Acta 1458, 356–363.
Girvin, M. E., Rastogi, V. K., Abildgaard, F., Markley, J. L., and Fillingame, R. H. (1998). Biochemistry 37, 8817–8824.
Greie, J.-C., Deckers-Hebestreit, G., and Altendorf, K. (2000). Eur. J. Biochem. 267, 3040–3048.
Howitt, S. M., Rodgers, A. J. W., Jeffrey, P. D., and Cox, G. B. (1996). J. Biol. Chem. 271, 7038–7042.
Kersten, M. V., Dunn, S. D., Wise, J. G., and Vogel, P. D. (2000). Biochemistry 39, 3856–3860.
Kuo, P. H., Ketchum, C. J., and Nakamoto, R. K. (1998). FEBS Lett. 426, 217–220.
Lau, S. Y. M., Taneja, A. K., and Hodges, R. S. (1984). J. Biol. Chem. 259, 13253–13261.
McCormick, K. A., Deckers-Hebestreit, G., Altendorf, K., and Cain, B. D. (1993). J. Biol. Chem. 268, 24683–24691.
McLachlin, D. T. and Dunn, S. D. (1997). J. Biol. Chem. 272, 21233–21239.
McLachlin, D. T. and Dunn, S. D. (2000). Biochemistry 39, 3486–3490.
McLachlin, D. T., Bestard, J. A., and Dunn, S. D. (1998). J. Biol. Chem. 273, 15162–15168.
McLachlin, D. T., Coveny, A. M., Clark, S. M., and Dunn, S. D. (2000). J. Biol. Chem. 267, 7630–7636.
Monticello, R. A. and Brusilow, W. S. A. (1994). J. Bacteriol. 176, 1383–1389.
Nakamoto, R. K., Ketchum, C. J., and Al-Shawi, M. K. (1999). Annu. Rev. Biophys. Biomol. Struct. 28, 205–234.
Oster, G. and Wang, H. (2000). Biochim. Biophys. Acta 1458, 482–510.
Revington, M., Dunn, S. D., and Shaw, G. S., manuscript in preparation.
Revington, M., McLachlin, D. T., Shaw, G. S., and Dunn, S. D. (1999). J. Biol. Chem. 274, 31094–31101.
Rodgers, A. J. W. and Capaldi, R. A. (1998). J. Biol. Chem. 273, 29406–29410.
Rodgers, A. J. W., Wilkens, S., Aggeler, R., Morris, M. B., Howitt, S. M., and Capaldi, R. A. (1997). J. Biol. Chem. 272, 31058–31064.
Sawada, K., Kuroda, N., Watanabe, H., Moritani-Otsuka, C., and Kanazawa, H. (1997). J. Biol. Chem. 272, 30047–30053.
Shilton, B. H. and Dunn, S. D. (2000) unpublished observations.
Shilton, B. H., Revington, M., and Dunn, S. D. (1999) unpublished observations.
Sorgen, P. L., Bubb, M. R., McCormick, K. A., Edison, A. S., and Cain, B. D. (1998a). Biochemistry 37, 923–932.
Sorgen, P. L., Caviston, T. L., Perry, R. C., and Cain, B. D. (1998b). J. Biol. Chem. 273, 27873–27878.
Sorgen, P. L., Bubb, M. R., and Cain, B. D. (1999). J. Biol Chem. 274, 36261–36266.
Soubannier, V., Rusconi, F., Vaillier, J., Arselin, G., Chaignepain, S., Graves, P.-V., Schmitter, J.-M., Zhang, J. L., Mueller, D., and Velours, J. (1999). Biochemistry, 38, 15017–15024.
Stock, D., Leslie, A. G. W., and Walker, J. E. (1999). Science 286, 1700–1705.
Takeyama, M., Noumi, T., Maeda, M., and Futai, M. (1988). J. Biol. Chem. 263, 16106–16112.
Tiburzy, H. J. and Berzborn, R. J. (1997). Z. Naturforsch 52c, 789–798.
Uhlin, U., Cox, G. B., and Guss, J. M. (1997). Structure 5, 1219–1230.
Walker, J. E. (ed.) (2000). Special issue: The Mechanisms of F1F0-ATPase. Amsterdam: Elsevier Science, [290 pp.] (Biochim. Biophys. Acta. 1458, Nos. 2–3).
Weber, J. and Senior, A. E. (1997). Biochim. Biophys. Acta 1319, 19–58.
Wilkens, S., Dahlquist, F. W., McIntosh, L. P., Donaldson, L. W., and Capaldi, R. A. (1995). Natur. Struct. Biol. 2, 961–967.
Wilkens, S., Dunn, S. D., Chandler, J., Dahlquist, F.W., and Capaldi, R. A. (1997). Natur. Struct. Biol. 4, 198–201.
Wilkens, S., Zhou, J., Nakayama, R., Dunn, S. D., and Capaldi, R. A. (2000). J. Mol. Biol. 295, 387–391.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dunn, S.D., Revington, M., Cipriano, D.J. et al. The b Subunit of Escherichia coli ATP Synthase . J Bioenerg Biomembr 32, 347–355 (2000). https://doi.org/10.1023/A:1005571818730
Issue Date:
DOI: https://doi.org/10.1023/A:1005571818730