Abstract
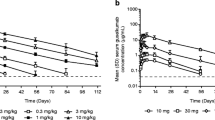
The pharmacokinetics of hu1124, a human anti-CD11a antibody, were investigated in human subjects with psoriasis. CD11a is a subunit of LFA-1, a cell surface molecule involved in T cell mediated immune responses. Subjects received a single dose of 0.03, 0.1, 0.3, 0.6, 1, 2, 3, or 10 mg/kg of hu1124 intravenously over 1–3 hr. Blood samples were collected at selected times from 60 min to 72 days after administration. Plasma samples were assayed for hu1124 by ELISA, and pharmacokinetic analyses were performed on the drug plasma concentrations. As the dose of hu1124 was increased, the clearance decreased from 322 ml/day per kg at 0.1 mg/kg to 6.6 ml/day per kg at 10 mg/kg of hu1124. The plasma hu1124 concentration–time profile suggested that the clearance of hu1124 was saturable above 10 μg/ml. In addition, treatment with hu1124 caused a rapid reduction in the level of CD11a expression on CD3-positive lymphocytes (T cells) to about 25% of pretreatment levels. Regardless of the hu1124 dose administered, cell surface CD11a remained at this reduced level as long as hu1124 was detectable (>0.025 μg/ml) in the plasma. When hu1124 levels fell below 3 μg/ml, the drug was rapidly cleared from the circulation and expression of CD11a returned to normal within 7–10 days thereafter. In vitro, half-maximal binding of hu1124 to lymphocytes was achieved at about 0.1 μg/ml and saturation required more than 10 μg/ml. One of the receptor-mediated pharmacokinetic/pharmacodynamic models which was developed describes the dynamic interaction of hu1124 binding to CD11a, resulting in the removal of hu1124 from the circulation and reduction of cell surface CD11a. The model accounts for the continually changing number of CD11a molecules available for removing hu1124 from the circulation based on prior exposure of cells expressing CD11a to hu1124. In addition, the model also accounts for saturation of CD11a molecules by hu1124 at drug concentrations of approximately 10 μg/ml, thereby reducing the clearance rate of hu1124 with increasing dose.
Similar content being viewed by others
REFERENCES
C. Dubey and M. Croft. Accessory molecule recognition of naive CD4 T cell activation. Immunol. Res. 15:114–125 (1996).
J. E. K. Hildreth, F. M. Gotch, P. D. K. Hildreth, and A. J. McMichael. A human lymphocyte-associated antigen involved in cell-mediated lympholysis. Eur. J. Immunol. 13:202–208 (1983).
A. Fischer, A. Durandy, G. Sterkers, and C. Griscelli. Role of the LFA-1 molecule in cellular interactions required for antibody production in humans. J. Immunol. 136:3198–3203 (1986).
J. E. K. Hildreth and J. T. August. The human lymphocyte function-associated (HLFA) antigen and a related macrophage differentiation antigen (HMac-1): Functional effects of subunit-specific monoclonal antibodies. J. Immunol. 134:3272–3280 (1985).
M. L. Dustin and T. A. Springer. LFA-1 interaction with ICAM-1 is one of at least three mechanisms for lymphocyte adhesion to cultured endothelial cells. J. Cell Biol. 107:321 (1988).
W. A. Werther, T. N. Gonzalez, S. J. O'Connor, S. McCabe, B. Chan, T. Hotaling, M. Champe, J. A. Fox, P. M. Jardieu, P. W. Berman, and L. G. Presta. Humanization of an anti-lymphocyte function-associated antigen (LFA)-1 monoclonal antibody and reengineering of the humanized antibody for binding to rhesus LFA-1. J. Immunol. 157:4986–4995 (1996).
S. L. Gottlieb, P. Gilleaudeau, R. Johnson, L. Estes, T. G. Woodworth, A. B. Gottlieb, and J. G. Krueger. Response of psoriasis to a lymphocyte-selective toxin (DAB389IL-2) suggests a primary immune, but not keratinocyte, pathogenic basis. Nature Med. 1:442–447 (1995).
W. Krzyzanski and W. J. Jusko. Mathematical formalism and characteristics of four basic models of indirect pharmacodynamic responses for drug infusions. J. Pharmacokin. and Biopharm. 26:385–408 (1998).
D. Z. D'Argenio and A. Schumitzky. ADAPT II User's Guide, Release 4. Biomedical Resource, U. Southern California, Los Angeles, 1997.
A. J. Boeckmann, L. B. Sheiner, and S. L. Beal. NONMEM V User's Help Guide. NONMEM Project Group, U. California at San Francisco, 1998.
M. O. Karlsson, E. N. Jonsson, C. G. Wiltse, and J. R. Wade. Assumption testing in population pharmacokinetic models: illustrated with an analysis of moxonidine data from congestive heart failure patients. J. Pharmacokin. Biopharm. 26:207–246 (1998).
W. A. Colburn. Specific antibodies and Fab fragments to alter the pharmacokinetics and reverse the pharmacologic/toxicologic effects of drugs. Drug Metab. Rev. 11:223–262 (1980).
T. A. Waldmann and W. Strober. Metabolism of immunoglobulins. Prog. Allergy 13:1–110 (1969).
T. A. Waldmann, W. Strober, and R. M. Blaese. Variations in the metabolism of immunoglobulins measured by turnover rates. In E. Merler (ed.), Immunoglobulins-Biological Aspects and Uses, National Academy of Sciences, Washington, DC, 1970, pp. 33–51.
J. S. Sancho, E. Gonzalez, J. F. Escanero, and J. Egido. Binding kinetics of monomeric and aggregated IgG to Kupffer cells and hepatocytes of mice. Immunology 53:283–289 (1984).
N. R. Srinivas, R. S. Weiner, W. C. Shyu, J. D. Calore, D. Trischler, L. K. Tay, J. S. Lee, D. S. Greene, and R. H. Barbhaiya. A pharmacokinetic study of intravenous CTLA4Ig, a novel immunosuppressive agent, in mice. J. Pharm. Sci. 85:296–298 (1996).
D. M. Sheeley, B. M. Merrill, and L. C. E. Taylor. Characterization of monoclonal antibody glycosylation: Comparison of expression systems and identification of terminal α-linked galactose. Anal. Biochem. 247:102–110 (1997).
F. H. Routier, M. J. Davies, K. Bergemann, and E. F. Hounsell. The glycosylation pattern of a humanized IgGl antibody (D1.3) expressed in CHO cells. Glyconjugate J. 14:201–207 (1997).
P. S. Linsley, W. Brady, M. Urnes, L. S. Grosmaire, N. K. Damle, and J. A. Ledbetter. CTLA-4 is a second receptor for the B cell activation antigen B7. J. Exp. Med. 174:561–569 (1991).
P. S. Linsley, P. M. Wallace, J. Johnson, M. G. Gibson, J. L. Greene, J. A. Ledbetter, C. Singh, and M. A. Tepper. Immunosuppression in vivo by a soluble form of the CTLA-4 T cell activation molecule. Science 257:792–795 (1992).
N. R. Srinivas, R. S. Weiner, G. Warner, W. C. Shyu, T. Davidson, C. G. Fadrowski, L. K. Tay, J. S. Lee, D. S. Greene, and R. H. Barbhaiya. Pharmacokinetics and pharmacodynamics of CTLA4Ig (BMS-188667), a novel immunosuppressive agent, in monkeys following multiple doses. J. Pharm. Sci. 85:1–4 (1996).
P. K. T. Li, M. G. Nicholls, and K. N. Lai. The complications of newer transplant antirejection drugs: treatment with cyclosporin A, OKT3, and FK506. Adverse Drug React. Acute Poisoning Rev. 9:123–155 (1990).
G. Goldstein, J. N. Douglas, K. R. Henell, and I. L. Smith. Pharmacokinetic study of Orthoclone OKT3 serum levels during treatment of acute renal allograft rejection. Transplantation 46:587–589 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bauer, R.J., Dedrick, R.L., White, M.L. et al. Population Pharmacokinetics and Pharmacodynamics of the Anti-CD11a Antibody hu1124 in Human Subjects with Psoriasis. J Pharmacokinet Pharmacodyn 27, 397–420 (1999). https://doi.org/10.1023/A:1020917122093
Published:
Issue Date:
DOI: https://doi.org/10.1023/A:1020917122093