Abstract
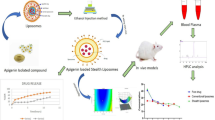
Purpose. The aim of the study was to prepare stealth and remoteloading proliposome (SRP-L) to carry doxorubicin (DXR) and evaluatethe pharmacokinetics, acute toxicity, and anticancer effect of DXRcarried with SRP-L.
Methods. SRP-L was transparent solution. When SRP-L was injectedinto 0.9% NaCl aqueous solution containing DXR, liposomes formedand automatically loaded DXR (SRP-L-DXR). The long circulation ofSRP-L-DXR was evaluated using the pharmacokinetics ofSRP-L-DXR, cardiolipin liposomal DXR (CL-DXR) and free DXR (F-DXR).The acute toxicity and anticancer effect of SRP-L-DXR were evaluatedin C57BL/6 mice and murine hystocytoma M5076 tumor model.
Results. The average diameter of SRP-L-DXR in pure water was112.9 ± 8.6 (nm) and the encapsulation efficiency of SRP-L-DXRwas 96.5 ± 0.2% in pure water, 95.5 ± 0.1% in 5% glucose and 98.01± 0.6% in 0.9% NaCl. The plasma concentration of SRP-L-DXR wasmuch higher than those of F-DXR and CL-DXR. Compared with thatof F-DXR, the SRP-L-DXR had lower acute toxicity and its anticancereffects depended upon the therapeutic treatment.
Conclusions. A novel proliposome (SRP-L) was developed, whichcould automatically load DXR and form SRP-L-DXR with excellentcharacteristics. SRP-L-DXR had lower acute toxicity but was notalways more effective for the treatment of the ascitic M5076 thanF-DXR.
Similar content being viewed by others
REFERENCES
E. A. Fossen and Z. A. Tokes. In vitro and in vivo studies with adriamycin liposomes. Biochem. Biophys. Res. Commun. 91:1295–1298 (1979).
A. D. Bangham, M. M. Standout, and J. C. Watkins. Diffusion of univalent ions across the lamellae of swollen phospholipids. J. Mol. Bio. 13:238–252 (1965).
N. Z. Wu, Da. D. Ruddoll, T. L. Needham, R. Whorton, and M. W. Dewhirst. Increased microvascular permeability contributes to preferential accumulation of STEALTH liposomes in tumor tissue. Cancer Res. 53:3765–3770 (1993).
J. Vaage, D. Donovan, P. Uster, and P. Working. Tumor uptake of doxorubicin in polyethylene-glyco coated liposomes and therapeutic effect against a xenografted human pancreatic carcinoma. Br. J. Cancer 75:482–488 (1997).
M. Mori, A. A. Klibanove, V. P. Torchilin, and L. Huang. Influence of the steric barrier activity of amphipathic poly (ethyleneglyco) and ganglioside GM1 on the circulation time of liposomes and on the target binding of immunoliposomes in vivo. FEBS. LETTERS 284:63–265 (1991).
D. D. Lasic, P. M. Frederick, M. C. Stuart, Y. Barenholz, and T. J. Mcintosh. Gelation of liposome interior: A novel method for drug encapsulation. FEBS. Lett. 312:255–258 (1992).
Y. Barenholz and G. Haren. Method of amphiphilic drugs loading into liposomes by pH gradient method. US patent 5, 192, 549 (1993).
P. C. Gokhale, B. Radhakrishnan, S. R. Husain, D. R. Abernethy, R. Sacher, A. Dristschilo, and A. Rahman. An improved method of encapsulation of doxorubicin in liposomes: pharmacological, toxicological and therapeutic evaluation. Br. J. Cancer 74:43–48 (1996).
G. Delgado, R. K. Potkul, J. A. Treat, G. S. Lewandowski, J. F. Barter, D. Forst, and A. Rahman. A phase I/II study of intraperitoneally administered doxorubicin entrapped in cardiolipin liposomes in patients with ovarian cancer. Am. J. Obstet. Gynecol. 160:812–817 (1989).
S. Batzri and E. D. Korn. Single bilayer liposomes prepared without sonication. Biochim. Biophys. Acta. 298:1015–1019 (1973).
A. Rahman, D. Carmichael, M. Harris, and J. K. Roh. Comparative pharmacokinetics of free doxorubicin and doxorubicin entrapped in cardiolipin liposomes. Cancer Res. 46:2295–2299 (1986).
S. K. Cox, A. V. Wilke, and D. Frazier. Determination of adriamycin in plasma and tissue biospices. J. Chromatogr. 564:322–329 (1991).
K. Yamaoka, Y. Tanigawara, T. Nakagawa, and T. Uno. A pharmacokinetic analysis program (multi) for microcomputer. J. Pharmacobiodyn. 4:879–885 (1981).
N. I Payney, I. Browning, and C. A. Hynes. Characterization of proliposome. J. Pharm. Sci. 75:330–333 (1986).
S. M. Johnson. The effect of charge and cholesterol on the size and thickness of sonicated phospholipid vesicles. Biochim. Biophys. Acta 307:27–41 (1973).
R. L. Hamilton, J. Goreke, L. S. S. Guo, M. C. Williams, and R. J. Havel. Unilamellar liposomes made with the French pressure cell: a simple preparative and semiquantitative technique. J. Lipid. Res. 21:981–992 (1980).
F. Szoka, F. Olson, T. Health, W. Vail, E. Mayhew, and D. Papahajopoulos. Preparation of unilamellar liposomes of intermediate size (0.1¶ 0.2 µm) by a combination of reverse phase evaporation and extrusion through polycarbonate membranes. Biochim. Biophys. Acta 601:559–571 (1980).
J. Vaage, D. Donovan, E. Mayhew, P. Uster, and M. Woodle. Therapy of mouse mammary carcinomas with vincristine and doxorubicin encapsulated in sterically stabilized liposomes. Int. J. Cancer 54:959–964 (1993).
J. P. Wang, D. S. Su, and X. Q. Gu. Studies on acute toxicity and antitumor effects of liposomal adriamycin. Chinese Pharmaceutical Journal 33:658–660 (1998).
R. K. Jain. Barriers to drug delivery in solid tumors. Scientific American 271:58–65 (1994).
A. Rahman, A. Fumagalli, B. Barbieri, P. S. Schein, and A.M. Casazza: Antitumor and toxicity evaluation of free doxorubicin and doxorubicin entrapped in cardiolipin liposomes. Cancer Chemother. Pharmacol. 16:22–27 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wang, J.P., Maitani, Y., Takayama, K. et al. Pharmacokinetics and Antitumor Effect of Doxorubicin Carried by Stealth and Remote Loading Proliposome. Pharm Res 17, 782–787 (2000). https://doi.org/10.1023/A:1007543805947
Issue Date:
DOI: https://doi.org/10.1023/A:1007543805947