Summary
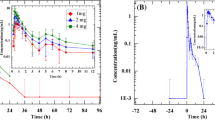
Six healthy volunteers received single 20-mg intravenous (IV) and 80-mg oral doses of propranolol on two occasions in random sequence. Serum propranolol concentrations were determined by gas chromatography in multiple samples drawn during 24 h after each dose. Mean (±SE) kinetic variables for IV propranolol were: elimination half-life (t1/2β), 5.3 (±0.6) h; volume of distribution, 2.3 (±0.3) l/kg; total clearance, 4.9 (±0.3) ml/min/kg; predicted extraction ratio, 0.23 (±0.02). After single oral doses, t1/2β (3.8±0.2 h) tended to be smaller than after the IV dose, and actual systemic availability (0.60±0.07) was less than that based on the predicted extraction ratio. During multiple oral dosage (80 mg every 12 h), observed steady state serum levels (47±5 ng/ml) tended to be less than those predicted based on the single oral dose (61±5 ng/ml), thus providing no evidence for reduced propranolol clearance at steady-state. Echocardiographic measurements of left ventricular performance (posterior wall velocity, diastolic dimensions) made during the single-dose oral study indicated significant impairment of function; impairment was maximal at 3 h post-dosage, and corresponded to the time of the peak serum propranolol concentration (341 ng/ml).
Zusammenfassung
Sechs gesunde Probanden erhielten in randomisierter Reihenfolge 20 mg Propranolol i.v. und 80 mg oral. Die Serumkonzentrationen an Propranolol wurden gaschromatographische über 24 h nach jeder Dosis bestimmt. Die kinetischen Variablen für Propranolol (±SE) lauten: Eliminationshalbwertzeit (t1/2β):5,3 h±0,6 h; Verteilungsvolumen: 2,3±0,3 l/kg; totale Clearance 4,9±0,3 ml/min/kg; berechnete Extraktionsrate: 0,23±0,02. Nach oraler Propranololgabe war t1/2β mit 3,8±0,2 h kürzer als nach i.v. Applikation und die systemische Verfügbarkeit mit 0,60±0,07 geringfügig niedriger als von der Extraktionsrate zu erwarten. Die steady state Serumkonzentrationen unter Mehrfachgabe (80 mg alle 12 h) lagen mit 47±5 ng/ml niedriger als die nach den Ergebnissen der Einmaldosis berechneten Spiegel (61±5 ng/ml). Die echokardiographisch bestimmten linksventrikulären Funktionsparameter (Kontraktionsgeschwindigkeit der Hinterwand, enddiastolischer Durchmesser), die unter der einmaligen 80 mg Dosis bestimmt wurden, zeigten eine Beeinträchtigung der Ventrikelfunktion mit einem Maximum 3 h nach Propranolol-Applikation. Dieser Zeitpunkt korrelierte mit den maximalen Serumkonzentrationen (341 ng/ml).
Similar content being viewed by others
References
Nies AS, Shand DG (1975) Clinical pharmacology of propranolol. Circulation 52:6–15
Johnson G, Regardh CG (1976) Clinical pharmacokinetics of β-adrenoreceptor blocking drugs. Clin Pharmacokinet 1:233–263
Shand DG (1974) Pharmacokinetic properties of the β-adrenergic receptor blocking drugs. Drugs 7:39–47
Evans GH, Shand DG (1973) Disposition of propranolol. V. Drug accumulation and steady-state concentrations during chronic oral administration in man. Clin Pharmacol Ther 14:487–493
Wood AJJ, Carr K, Vestal RE, Belcher S, Wilkinson GR, Shand DG (1978) Direct measurement of propranolol bioavailability during accumulation to steady-state. Br J Clin Pharmacol 6:345–350
Chidsey CA, Morselli PL, Bianchetti G, Morganiti A, Leonetti G, Zanchetti A (1975) Studies on the absorption and removal of propranolol in hypertensive patients during therapy. Circulation 52:313–318
Ochs HR, Grube E, Greenblatt DJ, Woo E, Bodem G (1980) Intravenous quinidine: pharmacokinetic properties and effects on left ventricular performance in humans. Am Heart J 99:468–475
DiSalle E, Baker KM, Bareggi SR, Watkins WD, Chidsey CA, Frigerio A, Morselli PL (1973) A sensitive gas chromatographic method for the determination of propranolol in human plasma. J Chromatogr 84:347–353
Ochs HR, Bodem G, Bales G, Greenblatt DJ, Smith TW (1978) Increased clearance of digoxin in rabbits during repeated administration. J Pharmacol Exp Ther 205:516–524
Greenblatt DJ (1979) Predicting steady-state serum concentrations of drugs. Ann Rev Pharmacol Toxicol 19:347–356
Gomeni R, Bianchetti G, Sega R, Morselli PL (1977) Pharmacokinetics of propranolol in normal healthy volunteers. J Pharmacokinet Biopharm 5:183–192
Kornhauser DM, Wood AJJ, Vestal RE, Wilkinson GR, Branch RA, Shand DG (1978) Biological determinants of propranolol disposition in man. Clin Pharmacol Ther 23:165–174
Crawford MH, Lindenfield JA, O'Rourke RA (1980) Effects of oral propranolol on left ventricular size and performance during exercise and acute pressure loading. Circulation 61:549–554
Kanto J, Kleimola T, Mantyla R, Syvalahti E (1976) Pharmacological effect of propranolol after a single oral dose. Acta Pharmacol Toxicol 39:573–576
Port S, Cobb FR, Jones RH (1980) Effects of propranolol on left ventricular function in normal men. Circulation 61:358–366
von Bibra H, Gibson DG, Nityanandan K (1980) Effects of propranolol on left ventricular wall movement in patients with ischaemic heart disease. Br Heart J 43:293–300
Cathcart-Rake WF, Coker JE, Atkins FL, Huffman DH, Hassanein KM, Shen DD, Azarnoff DL (1980) The effect of concurrent oral administration of propranolol and disopyramide on cardiac function in healthy men. Circulation 61:938–945
Gibson DG (1974) Pharmacodynamic properties of β-adrenergic receptor blocking drugs in man. Drugs 7:8–38
von Herick R, Aronow WS (1979) Effects of oxprenolol and propranolol on systolic time intervals. Clin Pharmacol Ther 24:678–682
Greenblatt DJ, Koch-Weser J (1974) Adverse reactions to beta adrenergic receptor blocking drugs: a report from the Boston Collaborative Drug Surveillance Program. Drugs 7:118–129
Author information
Authors and Affiliations
Additional information
Supported in part by Grant Oc 10/6-3 from Deutsche Forschungsgemeinschaft, Bonn-Bad Godesberg, FRG; and by Grant MH-34223 from the United States Public Health Service
Rights and permissions
About this article
Cite this article
Ochs, H.R., Grube, E., Greenblatt, D.J. et al. Kinetics and cardiac effects of propranolol in humans. Klin Wochenschr 60, 521–525 (1982). https://doi.org/10.1007/BF01756098
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01756098