Abstract
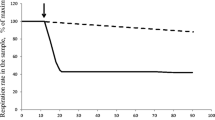
Degradation of microbiodies in the methanolutilizing yeastCandida boidinii was mainly studies by electron microscopical observation. The yeast cells precultured on methanol medium contained five to six microbodies per section and showed high activities of alcohol oxidase, catalase, formaldehyde dehydrogenase and formate dehydrogenase. When the precultured cells were transferred into an ethanol medium the number of microbodies and concomitantly the activities of alcohol oxidase and catalase decreased. After 6 h of cultivation microbodies were hardly detected. Also the activity of alcohol oxidase was not measurable and catalase activity was reduced to one tenth, whereas the activities of formaldehyde dehydrogenase and formate dehydrogenase decreased only to about 70%. Experiments with methanol-grown cells transferred into an ethanol medium without nitrogen source indicated that the inactivation of alcohol oxidase and catalase does not require protein synthesis. However, the reappearance of these enzymes is presumably due to de novo protein synthesis as shown by experiments with cycloheximide.
Similar content being viewed by others
References
Van Dijken, J. P., Veenhuis, M., Kreger-van Rij, N. J. W., Harder, W.: Microbodies in methanol-assimilating yeasts. Arch. Microbiol.102, 41–44 (1975)
Duntze, W., Neumann, D., Holzer, H.: Glucose induced inactivation of malate dehydrogenase in intact yeast cells. Eur. J. Biochem.3, 326–331 (1968)
Fukui, S., Tanaka, A., Kawamoto, S., Yasuhara, S., Teranishi, Y., Osumi, M.: Ultrastructure of methanol-utilizing yeast cells. Appearance of microbodies in relation to high catalase activity. J. Bacteriol.123, 317–328 (1975a)
Fukui, S., Kawamoto, S., Yasuhara, S., Tanaka, A., Osumi, M., Imaizumi, F.: Microbody of methanol-grown yeasts. Localisation of catalase and flavin-dependent alcohol oxidase in the isolated microbody. Eur. J. Biochem.59, 561–566 (1975b)
Gancedo, C.: Inactivation of fructose-1,6-diphosphatase by glucose in yeast. J. Bacteriol.107, 401–405 (1971)
Gancedo, C., Schwerzmann, K.: Inactivation by glucose of phosphoenolpyruvate carboxykinase fromSaccharomyces cerevisiae. Arch. Microbiol.109, 221–225 (1976)
Hake, T.: Studies on the reactions of OsO4 and KMnO4 with aminoacids, peptides, and proteins. Lab. Invest.14, 470–474 (1965)
Holzer, H.: Regulation of enzymes by enzyme catalyzed chemical modification, Adv. Enzymol.32, 297–326 (1969)
Holzer, H., Betz, H., Ebner, E.: Intracellular proteinases in microorganisms. In: Cur top. cell. regul. (B. L. Horecker, E. R. Stadtman, eds.), Vol. 9, pp. 103–156. New York: Academic Press 1975
Lowry, O. H., Rosebrough, N. J., Farr, J. G., Randall, R. J.: Protein measurement with the folin phenol reagent. J. Biol. Chem.193, 265–275 (1951)
Magni, G., Santarelli, J., Natalini, P., Ruggieri, S., Vita, A.: Catabolite inactivation of Bakers'-yeast uridine nucleosidase. Eur. J. Biochem.75, 77–82 (1977)
McClintock, D. K., Markus, G.: Conformational changes in aspartyltranscarbamylase. I. Proteolysis of the intact enzyme. J. Biol. Chem.243, 2855–2862 (1968)
Pine, M. J.: Response of intracellular proteolysis to alteration of bacterial protein and the implications in metabolic regulation. J. Bacteriol.93, 1527–1553 (1967)
Reynolds, E. S.: The use of lead citrate at high pH as an electronopaque stain in electron microscopy. J. Cell. Biol.17, 208–212 (1963)
Roggenkamp, R., Sahm, H., Hinkelmann, W., Wagner, F.: Alcohol oxidase and catalase in peroxisomes of methanol-grownCandida boidinii. Eur. J. Biochem.59, 231–236 (1975)
Roggenkamp, R., Sahm, H., Wagner, F.: Microbial assimilation of methanol. Induction and function of catalase inCandida boidinii. FEBS Lett.41, 283–286 (1974)
Sahm, H., Roggenkamp, R., Wagner, F., Hinkelmann, W.: Microbodies in methanol-grownCandida boidinii. J. Gen. Microbiol.88, 218–222 (1975)
Sahm, H., Wagner, F.: Mikrobielle Verwertung von Methanol. Isolierung und Charakterisierung der HefeCandida boidinii. Arch. Mikrobiol.84, 29–42 (1972)
Sahm, H., Wagner, F.: Microbial assimilation of methanol. Ethanol and methanol-oxidizing enzymes of the yeastCandida boidinii. Eur. J. Biochem.36, 250–256 (1973a)
Sahm, H., Wagner, F.: Mikrobielle Verwertung von Methanol. Eigenschaften der Formaldehyddehydrogenase und Formiatdehydrogenase ausCandida boidinii. Arch. Mikrobiol.90, 263–268 (1973b)
Schütte, H., Flossdorf, J., Sahm, H., Kula, M.-R.: Purification and properties of formaldehyde and formate dehydrogenase fromCandida bodinii. Eur. J. Biochem.62, 151–160 (1976)
Spurr, A. R.: A low-viscosity epoxy resin embedding medium for electron microscopy. J. Ultrastruct. Res.26, 31–46 (1969)
Tanaka, A., Yasuhara, S., Kawamoto, S., Fukui, S., Osumi, M.: Development of microbodies in the yeastKloeckera growing on methanol. J. Bacteriol.126, 919–927 (1976)
Tsubouchi, J., Tonomura, K., Tanaka, K.: Ultrastructure of microbodies of methanol-assimilating yeasts. J. Gen. Microbiol.22, 131–142 (1976)
Veenhuis, M., van Dijken, J. P., Harder, W.: Cytochemical studies on the localization of methanol oxidase and other oxidases in peroxisomes of methanol-grownHansenula polymorpha. Arch. Microbiol.111, 123–135 (1976)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bormann, C., Sahm, H. Degradation of microbodies in relation of activities of alcohol oxidase and catalase inCandida boidinii . Arch. Microbiol. 117, 67–72 (1978). https://doi.org/10.1007/BF00689353
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00689353