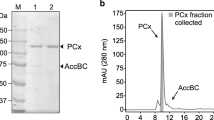
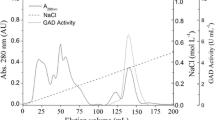
Abstract
Glutamate 1-semialdehyde aminotransferase has been separated from metabolically related activities by gel filtration and affinity chromatography. The enzyme was inhibited by gabaculin, 4-amino 5-fluoropentanoic acid and pyridoxal 5-phosphate and stimulated by pyridoxamine 5-phosphate. The activity of enzyme recovered by elution after electrophoresis in non-denaturing polyacrylamide gels was wholly dependent on pyridoxamine 5-phosphate. A mechanism for the enzyme-catalysed reaction based on these observations is discussed.
Similar content being viewed by others
Abbreviations
- AFPA:
-
4-amino 5-fluoropentanoic acid
- ALA:
-
δ-aminolaevulinic acid
- DTT:
-
dithiothreitol
- GSA:
-
glutamate 1-semialdehyde
- PAL-P:
-
pyridoxal 5-phosphate
- PAM-P:
-
pyridoxamine 5-phosphate
- PCC:
-
Paris Culture Collection
References
Avissar YJ, Beale SI (1989) Biosynthesis of tetrapyrrole pigment precursors. Pyridoxal requirement of the aminotransferase step in the formation of δ-aminolevulinate from glutamate in extracts of Chlorella vulgaris. Plant Physiol 89:852–859
Bull AD, Pakes JF, Hoult RC, Rogers LJ, Smith AJ (1989) Tetrapyrrole biosynthesis in a gabaculin tolerant mutant of Synechococcus 6301. Biochem Soc Trans 17:911–912
Castelfranco PA, Beale SI (1983) Chlorophyll biosynthesis: recent advances and areas of current interest. Annu Rev Plant Physiol 34:241–278
Gough SP, Kannangara CG, Bock K (1989) A new method for the synthesis of glutamate 1-semialdehyde. Characterization of its structure in solution by NMR spectroscopy. Carlsberg Res Commun 54:99–108
Grimm B, Bull A, Welinder KG, Gough SP, Kannangara CG (1989) Purification and partial amino acid sequence of glutamate 1-semialdehyde aminotransferase of barley and Synechococcus. Carlsberg Res Commun 54:67–79
Hendrick JL, Smith AJ (1968) Size and charge isomer separation and estimation of molecular weights of proteins by disc gel electrophoresis. Arch Biochem Biophys 126:155–164
Hoober JK, Kahn A, Ash DE, Gough S, Kannangara CG (1988) Biosynthesis of δ-aminolevulinate in greening barley leaves. XI. Structure of the substrate, mode of gabaculine inhibition, and the catalytic mechanism of glutamate 1-semialdehyde aminotransferase. Carlsberg Res Commun 53:11–25
Hoult RC, Rees D, Rogers LJ, Smith AJ (1986) Specific inhibition of tetrapyrrole biosynthesis by 3-amino 2,3-dihydrobenzoic acid (gabaculin) in the cyanobacterium Synechococcus 6301. Arch Microbiol 146:57–62
Kannangara CG, Gough SP (1978) Biosynthesis of δ-aminolevulinate in greening barley leaves: Glutamate 1-semialdehyde aminotransferase. Carlsberg Res Commun 43: 185–194
Kobayashi K, Miyazawa S, Endo A (1977) Isolation and inhibitory activity of gabaculine, a new potent inhibitor of γ-aminobutyrate aminotransferase produced by a streptomyces. FEBS Lett 76:207–210
Mau Y-HL, Wang W-Y (1988) Biosynthesis of δ-aminolevulinic acid in Chlamydomonas reinhardtii. Plant Physiol 86:793–797
Neville DM (1971) Molecular weight determination of protein-dodecyl sulphate complexes by gel electrophoresis in a discontinuous buffer system. J Biol Chem 246:6328–6334
Silverman RB, Levy MA (1980) Irreversible inactivation of γ-aminobutyric acid-α-ketoglutaric acid transaminase by 4-amino 5-halopentanoic acids. Biochem Biophys Res Commun 95: 250–255
Smith AJ, London J, Stanier RY (1967) Biochemical basis of obligate autotrophy in blue-green algae and thiobacilli. J Bacteriol 94:972–983
Smith AJ, Rogers LJ (1988) Tetrapyrrole biosynthesis — the C5 pathway. In: Rogers LJ, Gallon JR (eds) Proceedings of the Phytochemical Society of Europe. Biochemistry of the algae and cyanobacteria. Clarendon Press, Oxford, pp 69–96
Soper TS, Manning JM (1982) Inactivation of pyridoxal phosphate enzymes by gabaculine: correlation with enzymic exchange of β-protons. J Biol Chem 257:13930–13936
Wang W-Y, Gough SP, Kannangara CG (1981) Biosynthesis of δ-aminolevulinate in greening barley leaves IV. Isolation of three soluble enzymes required for the conversion of glutamate to δ-aminolevulinate. Carlsberg Res Commun 46:243–257
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bull, A.D., Breu, V., Kannangara, C.G. et al. Cyanobacterial glutamate 1-semialdehyde aminotransferase: requirement for pyridoxamine phosphate. Arch. Microbiol. 154, 56–59 (1990). https://doi.org/10.1007/BF00249178
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00249178