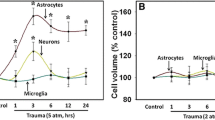
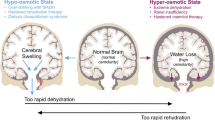
Summary
The ionic parameters of incubation media which foster both the development and subsequent reduction of swelling of slices of cerebral cortex under isosmotic conditions of incubation in vitro are described. A linear relationship between increasing chloride concentrations in incubation media and progressive swelling of tissue slices (under conditions of constant temperature and K+ concentrations and isotonicity of incubation media) is demonstrated. Subsequent reduction of chloride concentration in incubation media together with reciprocal replacement by isethionate is associated with significant and characteristic reduction in the volume of tissue swelling when all other conditions of incubation, including isotonicity of the media, are kept constant. The ionic composition of the fluid of swelling under different conditions of incubation is derived together with the ionic composition and expected transmembrane potentials of the neuronal compartment of cerebral cortex in vitro. Mechanisms involved in the development and subsequent reduction of swelling of cerebral cortex in vitro are discussed, and proposals for possible clinical applications are suggested.
Similar content being viewed by others
References
Ames, A., J.B. Isom and F.B. Nesbett: Effects of osmotic changes on water and electrolytes in nervous tissue. J. Physiol. (Lond.) 177, 246–262 (1965).
Bekaert, J., and G. Demeester: Influence of potassium concentration of the blood on the potassium level of the cerebrospinal fluid. Exp. Med. Surg. 12, 480–501 (1954).
Bourke, R.S.: Evidence for mediated transport of chloride in cat cerebral cortex in vitro. Exp. Brain Res. 8, 219–231 (1969).
—, E.S. Greenberg and D.B. Tower: Variation of cerebral cortex fluid spaces in vivo as a function of species brain size. Amer. J. Physiol. 208, 682–692 (1965).
—, and D.B. Tower: Fluid compartmentation and electrolytes of cat cerebral cortex in vitro. I. Swelling and solute distribution in mature cerebral cortex. J. Neurochem. 13, 1070–1094 (1966a).
—: Fluid compartmentation and electrolytes of cat cerebral cortex in vitro. Sodium, potassium and chloride of mature cerebral cortex. J. Neurochem. 13, 1099–1117 (1966b).
Conway, E.J., and H. Geoghegan: Molecular concentration of kidney cortex slices. J. Physiol. (Lond.) 130, 438–445 (1955).
Cserr, H.: Potassium exchange between cerebrospinal fluid, plasma, and brain. Amer. J. Physiol. 209, 1219–1226 (1965).
Cummins, J., and H. Hydén: Adenosine triphosphatase levels and adenosine triphosphatase in neurons, glia and neuronal membranes of the vestibular nucleus. Biochim. biophys. Acta (Amst.) 60, 271–283 (1962).
Davson, H., and J.F. Danielli: The Permeability of Natural Membranes, pp. 19–30. London: Cambridge University Press 1943.
Eccles, J.C.: The Physiology of Nerve Cells, p. 270. Baltimore: Johns Hopkins Press 1957.
Evans, J.P., and I.M. Scheinker: Histologic studies of the brain following head trauma. I. Post traumatic cerebral swelling and edema. J. Neurosurg. 2, 306–314 (1945).
Fencl, V., T.B. Miller and J.R. Pappenheimer: The respiratory response to disturbances of acid-base belance. In: Head Injury, pp. 414–430. Ed. by W. Caveness and A.E. Walker. Philadelphia: J.B. Lippincott Co. 1966.
Gibson, I.M., and H. McIlwain: Continuous recording of changes in membrane potential in mammalian cerebral tissues in vitro; recovery after depolarization by added substances. J. Physiol. (Lond.) 176, 261–263 (1965).
Hertz, L.: Neuroglial localization of potassium and sodium effects on respiration in brain. J. Neurochem. 13, 1373–1387 (1966).
—: Potassium effects on ion transport in brain slices. J. Neurochem. 15, 1–16 (1968).
Jenkner, F.L.: Über die anticholinerge Therapie der schweren geschlossenen Schädelhirntraumen. Langenbecks Arch. klin. Chir. 286, 91–98 (1957).
—: Treating and preventing cerebral edema. Acta neurol. lat.-amer. 4, 184–189 (1958).
Katzman, R.: Electrolyte distribution in mammalian central nervous system. Are glia high sodium cells? Neurology 11, 27–36 (1961).
Koch, A., J.B. Ranck and B.L. Newman: Ionic content of the neuroglia. Exp. Neurol. 6, 186–200 (1962).
Kuffler, S.W., and J.G. Nicholls: Glial cells in the central nervous system of the leech; their membrane potential and potassium content. Arch. exp. Path. Pharmakol. 248, 216–222 (1964).
— and R.K. Orkand: Physiological properties of glial cells in the central nervous system of amphibia. J. Neurophysiol. 29, 768–787 (1966).
—, and D.D. Potter: Glia in the leech central nervous system. Physiological properties and neuron-glia relationship. J. Neurophysiol. 27, 290–320 (1964).
Leaf, A.: On the mechanism of fluid exchange in tissues in vitro. Biochem. J. 62, 241–248 (1956).
Marshall, W.H.: Spreading depression of Leão. Physiol. Rev. 39, 239–279 (1959).
Nicholls, J.G., and S.W. Kuffler: Extracellular space as a pathway for exchange between blood and neurons in the central nervous system of the leech. Ionic composition of glial cells and neurons. J. Neurophysiol. 27, 645–671 (1964).
Orkand, R.K., J.G. Nicholls and S.W. Kuffler: Effect of nerve impulses on the membrane potential of glial cells in the central nervous system in amphibia. J. Neurophysiol. 29, 788–806 (1966).
Osterhout, J.V.: Transport of water from concentrated to dilute solutions in cells of Nitella. J. gen. Physiol. 32, 559–566 (1948).
Pappius, H.M., and K. A. C. Elliot: Water distribution in incubated slices of brain and other tissues. Canad. J. Biochem. 34, 1007–1022 (1956).
Rang, H.P., and J.M. Richie: On the electrogenic sodium pump in mammalian non-myelinated nerve fibres and its activation by various external cations. J. Physiol. (Lond.) 196, 183–221 (1968).
Stern, J.R., L.V. Eggleston, R. Hems and H.A. Krebs: Accumulation of glutamic acid in isolated brain tissue. Biochem. J. 44, 410–418 (1949).
Stewart-Wallace, A.M.: Biochemical studies of cerebral tissue and changes in cerebral edema. Brain 62, 426–438 (1939).
Tasaki, I., and J.J. Chang: Electric response of glia cells in cat brain. Science 128, 1209–1210 (1958).
Thomas, J., and H. McIlwain: Chloride content and metabolism of cerebral tissues in fluids low in chlorides. J. Neurochem. 1, 1–7 (1956).
Tobias, J.M.: A chemically specified molecular mechanism underlying excitation in nerve. A hypothesis. Nature (Lond.) 203, 13–17 (1964).
Tower, D.B.: Distribution of cerebral fluids and electrolytes in vivo and in vitro. In: Brain Edema, pp. 303–332. Ed. by F. Seitelberger and I. Klatzo. Vienna: Springer 1966.
Ussing, H.H.: The frog skin potential. J. gen. Physiol. 43, 135–147 (1960).
Van Harreveld, A.: Changes in volume of cortical neuronal elements during asphyxiation. Amer. J. Physiol. 191, 233–242 (1957).
—: Changes in diameter of apical dendrites during spreading depression. Amer. J. Physiol. 192, 457–463 (1958).
—: Brain Tissue Electrolytes, pp. 1–49. Washington: Butterworth, Inc. 1966.
Van Harreveld, and S. Ochs: Cerebral impedence changes after circulatory arrest. Amer. J. Physiol. 187, 180–192 (1956)
—, and J. P. Schadé: Chloride movements in cerebral cortex after circulatory arrest and during spreading depression. J. cell comp. Physiol. 54, 65–77 (1959).
—, and S. Tachibana: Recovery of cerebral cortex from asphyxiation. Amer. J. Physiol. 202, 59–65 (1962).
Varon, S., and H. McIlwain: Fluid content and compartments in isolated cerebral tissues. J. Neurochem. 8, 262–275 (1961).
Wallace, G.B., and B.B. Brodie: The distribution of iodide, thiocyanate, bromide and chloride in the central nervous system and spinal fluid. J. Pharmacol. exp. Ther. 65, 220–226 (1939).
White, J.C., J.R. Brooks, J.C. Goldthwaite and R.D. Adams: Changes in brain volume and blood content after experimental concussion. Ann. Surg. 118, 619–633 (1943).
Yannet, H.: Changes in the brain resulting from depletion of extracellular electrolytes. Amer. J. Physiol. 128, 683–689 (1940).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bourke, R.S. Studies of the development and subsequent reduction of swelling of mammalian cerebral cortex under isosmotic conditions in vitro . Exp Brain Res 8, 232–248 (1969). https://doi.org/10.1007/BF00234251
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00234251