Summary
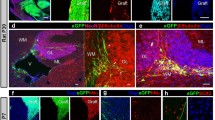
Using an immunocytochemical method, we examined the immunological responses of adult mice to intracerebellar syngeneic and allogeneic fetal mouse brainstem transplants (embryonic days 12–14). Syngeneic grafts and major histocompatibility complex (MHC)-compatible and non-MHC-incompatible allogeneic grafts survived well, showing no histological signs of rejection even 6 months after transplantation, and with no expression of MHC antigens being observed in any of the grafts. However, most cases of both MHC- and non-MHC-incompatible allografts showed rejection responses, such as marked neovascularization, cellular infiltration and necrosis, two weeks to one month after transplantation. In animals showing rejection, Class I MHC antigens were found on grafted neuronal tissue. An increased number of reactive astrocytes was also observed in the grafts. High levels of Class I antigen expression and prominent gliosis correlated with vigorous cellular infiltration. A quantitative analysis of T cell subsets in the animals showing rejection revealed that the L3T4/Lyt-2 ratio was 1.02±0.21 (mean ± S.D.), indicating that helper/inducer and cytotoxic/suppressor T cells appeared equally in the rejection of MHC- and non-MHC-incompatible allografts. We consider that in these experiments, the brain was not completely an immunologically privileged site, and that MHC- and non-MHC-incompatible intraparenchymal neural transplants were not shielded from host immune surveillance.
Similar content being viewed by others
References
Backlund E-O, Granberg P-O, Hamberger B, Knutsson E, Mårtensson A, Sedvall G, Seiger Å, Olson L (1985) Transplantation of adrenal medullary tissue to striatum in parkinsonism. First clinical trials. J Neurosurg 62: 169–173
Bakay RAE, Barrow DL, Fiandaca MS, Iuvone PM, Schiff A, Collins DC (1987) Biochemical and behavioral correction of MPTP parkinson-like syndrome by fetal cell transplantation. In: Azmitia EC, Björklund A (eds) Cell and tissue transplantation into the adult brain. Ann NY Acad Sci 495: 623–640
Barker CF, Billingham RE (1977) Immunologically privileged sites. Adv Immunol 25: 1–54
Björklund A, Segal M, Stenevi U (1979) Functional reinnervation of rat hippocampus by locus coeruleus implants. Brain Res 170: 409–426
Bradbury MWB, Westrop RJ (1983) Factors influencing exit of substances from cerebrospinal fluid into deep cervical lymph of the rabbit. J Physiol 339: 519–534
Brundin P, Nilsson OG, Gage FH, Björklund A (1985) Cyclosporin A increases survival of cross-species intrastriatal grafts of embryonic dopamine-containing neurons. Exp Brain Res 60: 204–208
Charlton HM, Barclay AN, Williams AF (1983) Detection of neuronal tissue from brain grafts with anti-Thy-1.1 antibody. Nature 305: 825–827
Fierz W, Endler B, Reske K, Wekerle H, Fontana A (1985) Astrocytes as antigen-presenting cells. I. Induction of Ia antigen expression on astrocytes by T cells via immune Interferon and its effect on antigen presentation. J Immunol 134: 3785–3793
Fontana A (1982) Astrocytes and lymphocytes: intercellular communication by growth factors. J Neurosci Res 8: 443–451
Fontana A, Fierz W, Wekerle H (1984) Astrocytes present myelin basic protein to encephalitogenic T-cell lines. Nature (Lond) 307: 273–276
Frank E, Pulver M, de Tribolet N (1986) Expression of class II major histocompatibility antigens on reactive astrocytes and endothelial cells within the gliosis surrounding metastases and abscesses. J Neuroimmunol 12: 29–36
Frei K, Bodmer S, Schwerdel C, Fontana A (1986) Astrocytederived interleukin 3 as a growth factor for microglia cells and peritoneal macrophages. J Immunol 137: 3521–3527
Gash D, Sladek JR, Sladek CD (1980) Functional development of grafted vasopressin neurons. Science 210: 1367–1369
Geyer SJ, Gill III TJ, Kunz HW, Moody E (1985) Immunogenetic aspects of transplantation in the rat brain. Transplantation 39: 244–247
Hall BM, Dorsch SE, Roser BJ (1978) The cellular basis of allograft rejection in vivo. I. The cellular requirements for first set rejection of heart grafts. J Exp Med 148: 878–889
Hall BM, Bishop GA, Farnsworth A, Duggin GG, Horvath JS, Sheil AGR, Tiller DJ (1984) Identification of the cellular subpopulations infiltrating rejecting cadaver renal allografts. Transplantation 37: 564–570
Hancock WW, Thomson NM, Atkins RC (1983) Composition of interstitial cellular infiltrate identified by monoclonal antibodies in renal biopsies of rejecting human renal allografts. Transplantation 35: 458–463
Head JR, Griffin WST (1985) Functional capacity of solid tissue transplants in the brain: evidence for immunological privilege. Proc R Soc Lond B 224: 375–387
Hsu S-M, Raine L, Fanger H (1981) Use of avidin-biotinperoxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem 29: 577–580
Inoue H, Kohsaka S, Yoshida K, Ohtani M, Toya S, Tsukada Y (1985) Cyclosporin A enhances the survivability of mouse cerebral cortex grafted into the third ventricle of rat brain. Neurosci Lett 54: 85–90
Lampson LA (1987) Molecular bases of the immune response to neural antigens. Trends Neurosci 10: 211–216
LeFrancois L, Bevan MJ (1984) A reexamination of the role of Lyt-2-positive T cells in murine skin graft rejection. J Exp Med 159: 57–67
Loveland BE, McKenzie IFC (1982) Which T cells cause graft rejection? Transplantation 33: 217–221
Madrazo I, Drucker-Colín R, Díaz V, Martínez-Mata J, Torres C, Becerril JJ (1987) Open microsurgical autograft of adrenal medulla to the right caudate nucleus in two patients with intractable Parkinson's disease. N Engl J Med 316: 831–834
Mason DW, Charlton HM, Jones AJ, Lavy CBD, Puklavec M, Simmonds SJ (1986) The fate of allogeneic and xenogeneic neuronal tissue transplanted into the third ventricle of rodents. Neuroscience 19: 685–694
McGeer PL, Itagaki S, Tago H, McGeer EG (1987) Reactive microglia in patients with senile dementia of the Alzheimer type are positive for the histocompatibility glycoprotein HLADR. Neurosci Lett 79: 195–200
Milton AD, Fabre JW (1985) Massive induction of donor-type class I and class II major histocompatibility complex antigens in rejecting cardiac allografts in the rat. J Exp Med 161: 98–112
Murphy JB, Sturm E (1923) Conditions determining the transplantability of tissue in the brain. J Exp Med 38: 183–197
Myrvik QN, Weiser RS (1984) Fundamentals of immunology, 2nd ed. Lea and Febiger, Philadelphia
Nicholas MK, Antel JP, Stefansson K, Arnason BGW (1987) Rejection of fetal neocortical neural transplants by H-2 incompatible mice. J Immunol 139: 2275–2283
Palmgren A (1948) A rapid method for selective silver staining of nerve fibers and nerve endings in mounted paraffin sections. Acta Zool 29: 377–392
Pepose JS, Gardner KM, Nestor MS, Foos RY, Pettit TH (1985) Detection of HLA class I and II antigens in rejected human corneal allografts. Ophthalmology 92: 1480–1484
Reif AE, Allen JM (1966) Mouse nervous tissue iso-antigens. Nature (Lond) 209: 523
Reif AE (1984) Transplantation of nerve tissue into brain. Appl Neurophysiol 47: 23–32
Sanfilippo F, Kolbeck PC, Vaughn WK, Bollinger RR (1985) Renal allograft cell infiltrates associated with irreversible rejection. Transplantation 40: 679–685
Strömberg I, Bygdeman M, Goldstein M, Seiger Å, Olson L (1986) Human fetal substantia nigra grafted to the dopaminedenervated striatum of immunosuppressed rats: evidence for functional reinnervation. Neurosci Lett 71: 271–276
Traugott U, Scheinberg LC, Raine CS (1985) On the presence of Ia-positive endothelial cells and astrocytes in multiple sclerosis lesions and its relevance to antigen presentation. J Neuroimmunol 8: 1–14
van Es A, Meyer CJLM, Oljans PJ, Tanke HJ, van Es LA (1984) Mononuclear cells in renal allografts. Transplantation 37: 134–139
Wong GHW, Bartlett PF, Clark-Lewis I, Battye F, Schrader JW (1984) Inducible expression of H-2 and Ia antigens on brain cells. Nature 310: 688–691
Wong GHW, Bartlett PF, Clark-Lewis I, McKimm-Breschkin JL, Schrader JW (1985) Interferon-γ induces the expression of H-2 and Ia antigens on brain cells. J Neuroimmunol 7: 255–278
Zhou C-F, Raisman G, Morris RJ (1985) Specific patterns of fibre outgrowth from transplants to host mice hippocampi, shown immunohistochemically by the use of allelic forms of Thy-1. Neuroscience 16: 819–833
Zinkernagel RM, Doherty PC (1979) MHC-restricted cytotoxic T cells: studies on the biological role of polymorphic major transplantation antigens determining T-cell restrictionspecificity, function, and responsiveness. Adv Immunol 27: 51–177
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Date, I., Kawamura, K. & Nakashima, H. Histological signs of immune reactions against allogeneic solid fetal neural grafts in the mouse cerebellum depend on the MHC locus. Exp Brain Res 73, 15–22 (1988). https://doi.org/10.1007/BF00279656
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00279656