Abstract
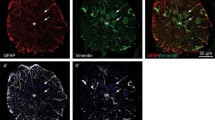
Exposure of the lumbar spinal cord of rats to X-rays 3 days after birth results in changes in the composition of central glia. Shortly after irradiation, there is both retardation of central myelin formation and a loss of integrity of the astrocyte-derived glia limitans on the dorsal surface of the cord. Subsequently, Schwann cells invade, undergo division and myelinate axons in the dorsal funiculi in the irradiated region of the cord, creating there an environment similar to that of peripheral nerve. The present study was undertaken to compare the ability of lesioned dorsal root axons to grow back into the altered glial environments that exist within the spinal cord after irradiation. This regrowth was assessed by injecting Fluoro-Gold into the spinal cord and subsequently examining neurons in the dorsal root ganglia (DRG) for the presence of this label. Numbers of retrogradely labeled neurons were counted in the DRG in both injured and contralateral non-injured sides. Non-irradiated control rats had almost no labeled DRG neurons on the injured side, whereas Fluoro-Gold labeled neurons were observed in substantial numbers in the DRG on the injured side of irradiated rats. There was a definite trend in the data, indicating that the longer the interval between irradiation and root injury, the greater the number of labeled neurons. Since the Fluoro-Gold labeling technique does not allow for visualization of the labeled axons within the spinal cord, a few animals were used to assess anterograde labeling with wheat germ agglutinin-conjugated horseradish peroxidase (WGA-HRP/HRP) from the dorsal root into the spinal cord. HRP-filled regenerating axons were visualized in dorsal white and gray matter of the irradiated spinal cord. Such axons were not present in the non-irradiated spinal cords. Radiation-induced changes in glial populations are discussed, particularly with regard to the temporal sequence of these changes and their possible relationship to the conversion of a normally non-permissive environment into one conducive to axonal regrowth.
Similar content being viewed by others
References
Aguayo AJ, David S, Bray G (1981) Influences of the glial environment on the elongation of axons after injury: transplantation studies in adult rodents. J Exp Biol 95:231–240
Ard MD, Bunge MB, Wood PM, Schachner M, Bunge RP (1991) Retinal neurite growth on astrocytes is not modified by extracellular matrix, anti-L1 antibody, or oligodendrocytes. Glia 4:70–82
Beal JA, Hall JL (1974) A light microscopic study of the effects of x-irradiation on the spinal cords of neonatal rats. J Neuropathol Exp Neurol 33:128–143
Bignami A, Chi NH, Dahl D (1984) Regenerating dorsal roots and the nerve entry zone. Immunofluorescence study with neurofilament and laminin antisera. Exp Neurol 85:426–436
Bignami A, Chi NH, Dahl D (1986) The role of neuroglia in axonal growth and regeneration. In: Wallace RB, Das GD (eds) Neural transplantation and regeneration. Springer, Berlin Heidelberg New York, pp 229–243
Bixby JL, Lilien J, Reichardt LF (1988) Identification of the major proteins that promote neuronal process outgrowth on Schwann cells in vitro. J Cell Biol 107:353–361
Blakemore WF, Patterson RC (1975) Observations on the interactions of Schwann cells and astrocytes following x-irradiation of neonatal rat spinal cord. J Neurocytol 4:573–585
Bologa L, Deugnier MA, Joubert R, Bisconte JC (1985) Myelin basic protein stimulates the proliferation of astrocytes: possible explanation for multiple sclerosis plaque formation. Brain Res 346:199–203
Cadelli DS, Bandtlow CE, Schwab ME (1992) Oligodendrocyteand myelin-associated inhibitors of neurite outgrowth: their involvement in the lack of CNS regeneration. Exp Neurol 115:189–192
Campenot RB (1982) Development of sympathetic neurons in compartmentalized cultures. I. Local control of neurite growth by nerve growth factor. Dev Biol 93:1–12
Carlstedt T (1988) Reinnervation of the mammalian spinal cord after neonatal dorsal root crush. J Neurocytol 17:335–350
Carlstedt T, Dalsgaard CJ, Molander C (1987) Regrowth of lesioned dorsal root nerve fibers into the spinal cord of neonatal rats. Neurosci Lett 74:14–18
Caroni P, Schwab ME (1988) Antibody to myelin-associated inhibitor for neurite growth neutralizes nonpermissive substrates of CNS white matter. Neuron 1:85–96
Dado RJ, Burstein R, Cliffer KD, Giesler GJ Jr (1990) Evidence that Fluoro-Gold can be transported avidly through fibers of passage. Brain Res 533:329–333
David S, Aguayo AJ (1981) Axonal elongation into peripheral nervous system ‘bridges’ after central nervous system injury in adult rats. Science 214:931–933
Fawcett JW, Rekos J, Bakst I (1989) Oligodendrocytes repel axons and cause growth cone collapse. J Cell Sci 92:93–100
Fawcett JW, Fersht W, Housden L, Schachner M, Pesheva P (1992) Axonal growth on astrocytes is not inhibited by oligodendrocytes. J Cell Sci 103:571–579
Gilmore SA (1963) The effects of x-irradiation on the spinal cords of neonatal rats. II. Histological observations. J Neuropathol Exp Neurol 22:294–301
Gilmore SA (1966) Delayed myelination induced by x-irradiation of the neonatal spinal cord. Neurology 16:749–753
Gilmore SA (1971) Autoradiographic studies of intramedullary Schwann cells in irradiated spinal cords of immature rats. Anat Rec 171:517–528
Gilmore SA, Duncan D (1968) On the presence of peripheral-like nervous and connective tissue within irradiated spinal cord. Anat Rec 160:675–690
Gilmore SA, Sims TJ (1986) The role of Schwann cells in the repair of glial cell deficits in the spinal cord. In: Wallace RB, Das GD (eds) Neural transplantation and regeneration. Springer, Berlin Heidelberg New York, pp 245–269
Gilmore SA, Sims TJ, Heard JK (1982) Autoradiographic and ultrastructural studies of areas of spinal cord occupied by Schwann cells and Schwann cell myelin. Brain Res 239:365–375
Gunderson RW, Barrett JN (1980) Characterization of the turning response of dorsal root neurites toward nerve growth factor. J Cell Biol 87:546–554
Guth L (1956) Regeneration in the mammalian peripheral nervous system. Physiol Rev 36:441–478
Guth L (1975) History of central nervous system regeneration research. Exp Neurol 48:3–15
Heard JK, Gilmore SA (1985) A comparison of histopathologic changes following x-irradiation of mid-thoracic and lumbosacral levels of neonatal rat spinal cord. Anat Rec 211:198–204
Heumann R, Korsching S, Bandtlow C, Thoenen H (1987) Changes of nerve growth factor synthesis in non-neuronal cells in response to sciatic nerve transection. J Cell Biol 104:1623–1631
Ide C, Tohyama K, Yokota R, Nitatori T, Onodera S (1983) Schwann cell basal lamina and nerve regeneration. Brain Res 288:61–75
Johnson EM Jr, Taniuchi M, Clark HB, Springer JE, Koh S, Tayrien MW, Loy R (1987) Demonstration of the retrograde transport of nerve growth factor receptor in the peripheral and central nervous system. J Neurosci 7:923–929
Levi-Montalcini R, Angeletti P (1968) Nerve growth factor. Physiol Rev 48:534–569
Leisi P (1985) Laminin-immunoreactive glia distinguish regenerative adult CNS systems from non-regenerative ones. EMBO J 4:2505–2511
Liuzzi FJ (1990) Proteolysis is a critical step in the physiological stop pathway: mechanisms involved in the blockade of axonal regeneration by mammalian astrocytes. Brain Res 512:277–283
Liuzzi FJ, Lasek RJ (1987) Astrocytes block axonal regeneration in mammals by activating the physiological stop pathway. Science 237:642–645
Manthorpe M, Engvall E, Ruoslahti F, Longo FM, Davis GE, Varon S (1983) Laminin promotes neuritic regeneration from cultured peripheral and central neurons. J Cell Biol 97:1882–1890
Maynard CW, Leonard RB, Coulter JD, Coggeshall RE (1977) Central connections of ventral root afferents as demonstrated by the HRP method. J Comp Neurol 172:601–608
Mesulam MM, Rosene DL (1979) Sensitivity in horseradish peroxidase neurohistochemistry: a comparative and quantatative study of nine methods. J Histochem Cytochem 27:763–773
Molander C, Grant G (1986) Laminar distribution and somatotopic organization of primary afferent fibers from hindlimb nerves in the dorsal horn. A study by transganglionic transport of horseradish peroxidase in the rat. Neuroscience 19:297–312
Reier PJ, Houle JD (1988) The glial scar: its bearing upon axonal elongation and transplantation approaches to CNS repair. In: Waxman SG (eds) Advances in Neurology, vol 47. Functional recovery in neurological disease, pp 87–138
Reier PJ, Stensaas LJ Guth L (1983) The astrocytic scar as an impediment to regeneration in the central nervous system. In: Kao CC, Bunge RP, Reier PJ (eds) Spinal cord reconstruction. Raven Press, New York, pp 163–195
Richardson PM, McGuinness UM, Aguayo AJ (1980) Axons from CNS neurons regenerate into PNS grafts. Nature 284:264–265
Richardson PM, McGuinness UM, Aguayo AJ (1982) Peripheral nerve autografts to the rat spinal cord: studies with axonal tracing methods. Brain Res 237:147–162
Richardson PM, Issa VMK, Aguayo AJ (1984) Regeneration of long spinal axons in the rat. J Neurocytol 13:165–182
Rivas R, Burmeister DW, Goldberg DJ (1992) Rapid effects of laminin on the growth cone. Neuron 8:107–115
Rivero-Melián C, Grant G (1990) Distribution of lumbar dorsal root fibers in the lower thoracic and lumbosacral spinal cord of the rat studied with choleragenoid horseradish peroxidase conjugate. J Comp Neurol 299:470–481
Rogers S, LeTourneau P, Palm S, McCarthy J, Furcht L (1983) Neurite extension by peripheral and central nervous system neurons in response to substratum-bound fibronectin and laminin. Dev Biol 98:212–220
Rudge JS, Silver J (1990) Inhibition of neurite outgrowth on astroglial scars in vitro. J Neurosci 10:3594–3603
Savio T, Schwab ME (1990) Lesioned corticospinal tract axons regenerate in myelin-free rat spinal cord. Proc Natl Acad Sci USA 87:4130–4133
Schnell L, Schwab ME (1990) Axonal regeneration in the rat spinal cord produced by an antibody against myelin-associated neurite growth inhibitors. Nature 343:269–272
Schwab ME, Caroni P (1988) Oligodendrocytes and CNS myelin are nonpermissive substrates for neurite growth and fibroblast spreading in vitro. J Neurosci 8:2381–2393
Schwab ME, Thoenen H (1985) Dissociated neurons regenerate into sciatic but not optic nerve expiants in culture irrespective of neurotrophic factors. J Neurosci 5:2415–2423
Schwab ME, Kapfhammer JP, Bandtlow CE (1993) Inhibitors of neurite growth. Ann Rev Neurosci 16:565–595
Silver J, Lorenz SE, Wahlsten D, Coughlin J (1982) Axonal guidance during development of the great cerebral commissures: descriptive and experimental studies, in vivo, on the role of preformed glial pathways. J Comp Neurol 210:10–29
Sims TJ, Gilmore SA (1983) Interactions between intraspinal Schwann cells and cellular constituents normally occurring in the spinal cord: an ultrastructural study in the irradiated rat. Brain Res 276:17–30
Sims TJ, Gilmore SA (1989) Interactions between Schwann cells and CNS axons following a delay in the normal formation of central myelin. Exp Brain Res 75:513–522
Sims TJ, Gilmore SA (1990) Regrowth of dorsal root axons into the spinal cord following irradiation. Neurosci Abstr 17:168
Sims TJ, Gilmore SA (1992) Glial response to dorsal root lesion in the irradiated spinal cord. Glia 6:96–107
Sims TJ, Gilmore SA, Waxman SG, Klinge E (1985) Dorsal-ventral differences in the glia limitans of the spinal cord: an ultrastructural study in developing normal and irradiated rats. J Neuropathol Exp Neurol 44:415–429
Smith GM, Miller RH, Silver J (1986) Changing role of forebrain astrocytes during development, regenerative failure and induced regeneration upon transplantation. J Comp Neurol 251:23–43
Sosale A, Robson JA, Stelzner DJ (1988) Laminin distribution during corticospinal tract development and after spinal cord injury. Exp Neurol 102:14–22
Stensaas LJ, Partlow LM, Burgess PR, Horch KW (1987) Inhibition of regeneration: the ultrastructure of reactive astrocytes and abortive axon terminals in the transition zone of the dorsal root. Prog Brain Res 71:457–468
Taniuchi M, Clark HB, Schweitzer JB, Johnson EM Jr (1988) Expression of nerve growth factor receptors by Schwann cells of axotomized peripheral nerves: ultrastructural location, suppression by axonal contact, and binding properties. J Neurosci 8:664–681
Thoenen H, Barde YA (1980) Physiology of nerve growth factor. Physiol Rev 60:1284–1335
Williams LR, Powell HC, Lundborg G, Varon S (1984) Competence of nerve tissue as distal insert promoting nerve regeneration in a silicone chamber. Brain Res 293:201–211
Windle WF (1956) Regeneration of axons in the vertebrate central nervous system. Physiol Rev 36:427–439
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sims, T.J., Gilmore, S.A. Regeneration of dorsal root axons into experimentally altered glial environments in the rat spinal cord. Exp Brain Res 99, 25–33 (1994). https://doi.org/10.1007/BF00241409
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00241409