Abstract
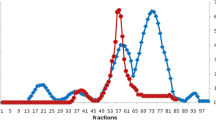
Two enzymes were purified from the green algaCodium fragile, collected in 1988 at Fukuoka, Japan, by batch-wise ion-exchange extraction, affinity chromatography and ion-exchange high-performance liquid chromatography (HPLC) using Boc-Ala-Ala-Pro-Arg-pNA as a substrate. The molecular weights of the enzymes were estimated as 38 000 and 39 000, using gel filtration HPLC. The enzymes had the same optimal pH range of 7 to 9 for both activities, and an exclusively hydrolyzed peptide bond on the carboxyl-terminal side of theL-arginine of peptidep-nitroanilides. The ratios of the enzymatic activity for X-Arg-pNA to X-Lys-pNA were larger than 100. The enzymes exhibited 30 times higher activity toward Boc-Ala-Ala-Pro-Arg-pNA when compared with trypsin. The activities were strongly inhibited by diisopropyl phosphofluoridate (DFP), and partially inhibited by phenylmethylsulfonyl fluoride (PMSF), benzamidine, leupeptin and antipain. The isolated enzymes were presumed to be trypsin-like serine protease from their primary substrate specificities and inactivation behavior.
Similar content being viewed by others
Literature cited
Boesman, M., Levy, M., Schenkein, I. (1976). Esteroproteolytic enzymes from the submaxillary gland. Archs Biochem. Biophys. 175: 463–476
Chase, T., Shaw, E. (1970). Titration of trypsin, plasmin, and thrombin with p-nitrophenyl p'-guanidinobenzoate HCl. Meth. Enzym. 19: 20–27
DelMar, E. G., Largman, C., Brondrick, J. W., Georkas, M. C. (1979). A sensitive new substrate for chymotrypsin. Analyt. Biochem. 99: 316–320
Erlanger, B. F., Kokowsky, N., Cohen, W. (1961). The preparation and properties of two new chromogenic substrates of trypsin. Archs. Biochem. Biophys. 95: 271–278
Gilles, A. M., Imhoff, J. M., Keil, B. (1979).α-Clostripain. J. biol. Chem. 254: 1462–1468
Graf, L., Jansco, A., Szilagyi, L., Hegyi, G., Pinter, K., Naray-Szabo, G., Heep, J., Medzihardszky, K., Rutter, W. J. (1988). Electrostatic complementarity within the substrate-binding pocket of trypsin. Proc. natl. Acad. Sci. U.S.A. 85: 4961–4965
Jameson, G. W., Roberts, D. V., Adams, R. W., Kyle, W. S. A., Elmore, D. T. (1973). Determination of the operational molarity of solutions of bovineα-chymotrypsin, trypsin, thrombin and factor Xa by spectrofluorimetric titration. Biochem. J. 31: 107–117
Kamahori, M., Yoshida, N., Tsuruyama, S., Tojo, A., Ogata, F., Makisumi, S. (1988). Chymotrypsin-like proteases in digestive juice of silkworm,Bombyx mori Isolation, characterization, and substrate specificity. Mem. Fac. Sci. Kyushu Univ. 16: 177–188
Kettner, C., Shaw, E. (1981). Inactivation of trypsin-like enzymes with peptides of arginine chloromethyl ketone. Meth. Enzym. 80: 826–842
Lottenberg, R., Christensen, U., Jackson, C. M., Coleman, P. L. (1981). Assay of coagulation proteases using peptide chromogenic and fluorogenic substrates. Meth. Enzym. 80: 341–369
Mitchell, W. M., Harrington, W. F. (1970). Clostripain. Meth. Enzym. 19: 635–642
Nishikata, M. (1984). Trypsin-like protease from soybean seeds. Purification and some properties. J. Biochem. 95: 1169–1177
Ochiai, Y., Katsuragi, T., Hashimoto, K. (1987). Proteins in three seaweeds, “Aosa”Ulva lactuca, “Arame”Eisenia bicyclis, and “Makusa”Gelidium amansii. Nippon Suisan Gakk. 53: 1051–1055
Schenkein, I., Levy, M., Franklin, E. C., Frangione, B. (1977). Proteolytic enzymes from the mouse submaxillary gland. Archs. Biochem. Biophys. 182: 64–70
Tsunematsu, H., Mizusaki, K., Makisumi, S., Okamoto, K., Tsunematsu, Y. (1985). A new serine protease which preferentially recognizesp-guanidino-L-phenylalanyl residue in ascitic plasma from ehrlich ascites tumor-bearing mice. Biochem. Biophys. Res. Commun. 128: 1233–1238
Williams, D. E., Reisfeld, R. A. (1964). Disc electrophoresis in polyacrylamide gels: Extension to new conditions of pH and buffer. Ann. N.Y. Acad. Sci. 121: 373–381
Yoshida, N., Tsuruyama, S., Nagata, K., Hirayama, K., Noda, K., Makisumi, S. (1988). Purification and characterization of an acidic amino acid specific endopeptidase ofStreptomyces griseus obtained from a commercial preparation (Pronase). J. Biochem. 104: 451–456
Zimmerman, M., Ashe, B. M. (1977). Substrate specificity of the elastase and the chymotrypsin-like enzyme of the human granulocyte. Biochim. Biophys. Acta 480: 241–245
Author information
Authors and Affiliations
Additional information
Communicated by M. Anraku, Tokyo
Rights and permissions
About this article
Cite this article
Kadokami, K., Yoshida, N., Mizusaki, K. et al. Some properties of trypsin-like proteases extracted from the seaweedCodium fragile and their purification. Mar. Biol. 107, 513–517 (1990). https://doi.org/10.1007/BF01313436
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01313436