Abstract
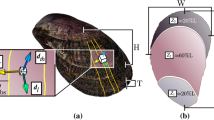
Shell microstructure and mantle behaviour relating to shell cementation was studied on adult oysters,Crassostrea gigas, Saccostrea mordax, andS. kegaki (collected from Shirahama, Wakayama Prefecture, Japan, in 1989 and 1990). At the place of cementation, the prismatic structure of the outermost shell layer is modified to a significant structure. This structure, named the ‘ridge-and-furrow structure’, consists of calcified ridges a few microns wide separated by furrows, both arranged parallel to the shell growth direction. The furrows are ultimately filled by shell material. The prismatic and ridge-and-furrow structures gradually merge in a transitional area where an intermediate type of the two structures occurs. The small size of the crystal units of the ridges and furrows is due to the close distribution of crystal seeds, especially close to pre-existing ridges. This is the basis of the difference between the ridge-and-furrow and the prismatic structures and also makes the former structure functional for cementation, in contrast to the latter. At the site of ongoing shell cementation, the mantle margin adpresses the shell margin onto the substrate. Experiments show that this pressing action is essential for cementation and probably also for the formation of the ridge-and-furrow structure. Even the right valve, which oysters nerve use to cement in natural conditions, forms the ridge-and-furrow structure and cements to the substrate if the pressing action of its mantle margin is induced under artificial conditions. Behavioural changes probably led oysters to switch from byssal attachment to cementation within a short time span when they acquired their cementing habit.
Similar content being viewed by others
References
Beedham, G. E. (1958a). Observation on the mantle of the Lamellibranchia. Q. J. microsc. Sci. 99: 181–197
Beedham, G. E. (1958b). Observations on the non-calcreous component of the shell of the Lamellibranchia. Q. J. microsc. Sci. 99: 341–357
Carriker, M. R., Palmer, E. P., Prezant, R. S. (1980). Functional ultramorphology of the dissoconch valve of the oysterCrassostrea virginica. Proc. natn. Shellfish. Ass. 70: 139–183
Carter, J. G. (1990). Skeletal biomineralization: patterns, processes and evolutionary trends. Van Nostrand Reinhold, New York
Carter, J. G., Clark, G. R. II (1985). Classification and phylogenetic significance of molluscan shell microstructure. In: Bottjer, D. J., Hickmann, C. S., Ward, P. D. (eds.) Mollusks, notes for a short course, 13. University of Tennessee, Department of Geological Science, Orlando, Florida, USA, p. 50–71
Cranfield, H. J. (1973a). A study of the morphology, ultrastructure, and histochemistry of the foot of the pediveliger ofOstrea edulis. Mar. Biol. 22: 187–202
Cranfield, H. J. (1973b). Observations on the behavior of the pediveliger ofOstrea edulis during attachment and cementing. Mar. Biol. 22: 203–209
Cranfield, H. J. (1973c). Observations on the function of the glands of the foot of the pediveliger ofOstrea edulis during settlement. Mar. Biol. 22: 210–223
Cranfield, H. J. (1974). Observations on the morphology of the mantle folds of the pediveliger ofOstrea edulis L. and their function during settlement. J. mar. biol. Ass. U.K. 54: 1–12
Cranfield, H. J. (1975). The ultrastructure and histochemistry of the larval cement ofOstrea edulis L. J. mar. biol. Ass. U.K. 55: 497–503
Grégoire, C. (1972) Structure of the molluscan shell. In: Florkin, M., Scheer, A. T. (eds.) Chemical zoology, Vol 7. Mollusca. Academic Press, New York, p. 45–102
Gruffydd, L. D., Lane, D. J. W., Beaumont, A. R. (1975). The glands of the larval foot inPecten maximus L. and possible homologues in other bivalves. J. mar. biol. Ass. U.K. 55: 463–476
Harper, E. M. (1992). Post-larval cementation in the Ostreidae and its implication for other cementing bivalvia. J. mollusc. Stud. 58: 37–47
Korringa, P. (1951). On the nature and function of “Chalky” deposits in the shell ofOstrea edulis. Proc. Calif. Acad. Sci. (Ser. 4) 27 (5): 133–158
Newell, N. D. (1960). The origin of the oysters. Proceedings of the International Paleontological Union, Part XXII, Report of 21th Session of International Geological Congress, Copenhagen, p. 81–86
Orton, J. H., Amirthalingam, C. (1927). Notes on shell-depositions in oysters. J. mar. biol. Ass. U.K. 14: 935–953
Petit, H., Davis, W., Jones, R. (1978). Morphological studies on the mantle of the fresh-water musselAmblema (Unionidae): scanning electron microscopy. Tissue Cell 104: 619–627
Purchon, R. D. (1968). The biology of the Mollusca. Pergamon Press, Oxford
Saleuddin, A. S. M., Petit, H. P. (1983). The mode of formation and the structure of periostracum. In: Saleuddin, A. S. M., Wilbur, K. M. (eds.) The Mollusca, Vol. 4. Physiology, Part 1. Academic Press, New York, p. 199–234
Stenzel, H. B. (1971). Oysters. In: R. C. Moore (ed.) Treatise on invertebrate paleontology, Part N. Mollusca 6, Bivalvia, Vol. 3. Geological Society of American Inc., Boulder, Colorado, p. 953–1224
Suzuki, S., Togo, Y., Hikida, Y. (1993). Using Meigen's staining for aragonite-calcite identification in fossil molluscan shells under the scanning electron microscope. J. geol. Soc. Japan 99: 1–7
Taylor, J. D., Kennedy, J. M., Hall, A. (1969). The shell structure and mineralogy of the Bivalvia-Introduction, Nucleacea-Trigonacea. Bull. Br. Mus. nat. Hist. (D. Zool.) 3: 1–125
Taylor, J. D., Kennedy, W. J. (1969). The influence of the periostracum on the shell structure of bivalve molluscs. Calcif. Tissue Res. 3: 274–283
Wilbur, K. M., Saleuddin, A. S. M. (1983). Shell formation. In: Wilbur, K. M. (ed.). The Mollusca, Vol. 4 Physiology, Part 1. Academic Press, New York, p. 235–287
Yonge, C. M. (1979). Cementation in bivalves. In: Van De Spoel, S., Van Bruggen, A. C., Lever, J. (eds.) Pathways in malacology. Bohn, Scheltema, Holkema, and Junk, Utrecht, p. 83–106
Author information
Authors and Affiliations
Additional information
Communicated by T. Ikeda, Nagasaki
Rights and permissions
About this article
Cite this article
Yamaguchi, K. Shell structure and behaviour related to cementation in oysters. Mar. Biol. 118, 89–100 (1994). https://doi.org/10.1007/BF00699222
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00699222