Summary
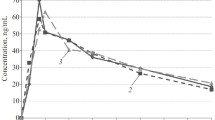
The pharmacokinetics of clonidine and its relation to blood pressure response and side effects were studied after single oral doses of 75 µg, 150 µg and 250 µg in normotensive subjects. Following oral administration, the drug was absorbed rapidly after an initial lag time of 19–22 min and peak levels were reached between 2.4 and 2.9 h. Sampling over 48 h was necessary for accurate estimation of pharmacokinetic parameters. Post-peak plasma concentration declined in a monoexponential manner and the half-life of the elimination phase ranged from 9.0 to 15.1 h. Maximum plasma concentration (Cmax) and area under curve (AUC) increased proportionally with increasing doses. Clonidine produced significant reductions in the pulse rate and a dose dependent decrease in blood pressure. Clonidine (150 µg) also produced significant reductions in plasma catecholamine levels.
Similar content being viewed by others
References
Berthelsen S, Pettinger WA (1977) A functional basis for classification of alpha-adrenergic receptors. Life Sci 21:595–606
Cho AK, Curry S (1969) The physiological disposition of 2-(2,6-dichlorophenylamine)-2-imidazoline. Biochem Pharmacol 18:511–520
Da Prada M, Zurcher G (1976) Simultaneous radioenzymatic determination of plasma and tissue adrenaline, noradrenaline and dopamine within the femtomole range. Life Sci 19:1161–1174
Davies DS, Wing LMH, Reid DM, Neill E, Tippett P, Dollery MB (1977) Pharmacokinetics and concentration effect relationships of intravenous and oral clonidine. Clin Pharmacol Ther 21 (5):593–601
Davies DS, Neill E, Reid JL (1978) Applications of deuterium labelling in pharmacokinetic and concentration effect studies of clonidine. In: Baillie TA (ed) Stable isotopes. Macmillan, London, pp 45–54
Dollery CT, Davies DS, Drafftan GH, Dragie HJ, Dean CR, Reid JL, Clare RA, Murray S (1975) The clinical pharmacology and pharmacokinetics of clonidine. Clin Pharmacol Ther 19:11–17
Frisk-Holmberg M, Edund PO, Paalzow L (1978) Pharmacokinetics of clonidine and its relation to the hypotensive effect in patients. Br J Clin Pharmacol 6:227–232
Gomeri R, Gomeri C (1979) AUTOMOD, a polyalgorithm for an integrated analysis of linear pharmacokinetic models. Comput Biol Med 9:39–48
Jarrott B, Spector S (1978) Disposition of clonidine in rats as determined by radioimmunoassay. J Pharmacol Exp Ther 207 (1):195–202
Louis WJ, Doyle AE, Anavekar SN, Johnston CI, Geffen LB, Rush R (1974) Plasma catecholamine, dopamine-beta-hydroxylase and renin levels in essential hypertension. Circ Res 34 (Suppl 1):57–63
Ng J, Phelan EL, McGregor DD, Laverty R, Taylor KM, Smirk H (1967) Properties of Catapress, a new hypotensive drug: A preliminary report. NZ Med J 66:864–870
Pettinger WA (1980) Pharmacology of clonidine. J Cardiovasc Pharmacol 2 (Suppl 1):521–528
Raftos J, Bauer GE, Lewis RG (1973) Clonidine in the treatment of severe hypertension. Med J Aust 1:786–793
Rehbinder D (1976) The metabolism of clonidine. In: Connolly ME (ed) Catapress in hypertension. Butterworth, London
Stewart A, Metz JB, Halter B, Porte D Jr, Paul Robertson R (1978) Suppression of plasma catecholamines and flushing by clonidine in man. J Clin Endocrinol Metab 46 (1):83–90
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Anavekar, S.N., Jarrott, B., Toscano, M. et al. Pharmacokinetic and pharmacodynamic studies of oral clonidine in normotensive subjects. Eur J Clin Pharmacol 23, 1–5 (1982). https://doi.org/10.1007/BF01061368
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01061368