Summary
The immunosuppressive activity of azathioprine (AZA) is unpredictable and depends on the formation of intracellular thiopurine ribonucleotides. However, the quantification of these active thiopurines presents difficult analytical problems. It has recently been postulated that plasma concentrations of 6-thiouric acid (6-TU) and 6-mercaptopurine (6-MP), metabolites of AZA, may provide more readily measurable indices of the pharmacologic activity of AZA. In order to evaluate the utility of 6-TU and 6-MP plasma concentrations in monitoring AZA therapy, we studied their pharmacokinetics in 6 renal transplant patients, and their in vitro immunosuppressive potency in a mixed lymphocyte proliferation assay.
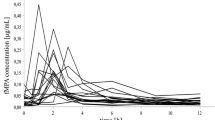
A peak plasma 6-TU concentration of 710.7 ng/ml was observed at 3.8 h after oral dosing. Good correlation was observed between the elimination t1/2 of 6-TU and serum creatinine, and between AUC over 24 h and serum creatinine. However, we did not observe a second peak in plasma 6-TU concentration that could be attributed to the degradation of active AZA metabolites. 6-MP plasma concentrations in the patients were low (mean peak concentration 36.0 ng/ml) and rapidly disappeared within 8 h. In vitro immunosuppressive activity could not be demonstrated for 6-TU over a concentration range of 1.25 ng/ml to 0.25 mg/ml.
We conclude that 6-TU is pharmacologically inert and is primarily eliminated by the kidneys. Our findings currently do not support the use of plasma concentrations of 6-TU or 6-MP to monitor AZA therapy. In order to optimize AZA therapy, analytical techniques that are technically feasible and that can directly quantify the active intracellular thiopurines are being explored.
Similar content being viewed by others
References
Bach JF (1975) Thiopurines. In: Newberger A, Tatum EL (eds) The mode of action of immunosuppressive agents. North-Holland. Amsterdam
Bach JF, Dardenne M (1971) The mechanism of azathioprine in renal failure. Transplantation 12: 253–259
Breter HJ, Zahn RK (1979) Quantification of intracellular metabolites of (35S)-6-mercaptopurine in L5178Y cells grown in time-course incubates. Cancer Res 39: 3744–3748
Canafax DM, Martel EJ, Ascher NL, Payne WD, Sutherland DER, Simmons RL, Najarian JS (1985) Two methods of managing cyclosporine nephrotoxicity: Conversion to azathioprine, prednisone, or cyclosporine, azathioprine, and prednisone. Transplant Proc 17: 1176–1177
Chan GLC, Canafax DM, Ascher NL, Dunn DL, Payne WD, Fryd DS, Noreen H, Sutherland DER, Simmons RL, Najarian JS (1988) HLA-identical sibling renal transplants: No rejections with a cyclosporine-azathioprine-prednisone protocol. Clin Transplantation 2: 9–14
Chan GLC, Canafax DM, Johnson CA (1987) The therapeutic use of azathioprine in renal transplantation. Pharmacotherapy 7: 165–177
Ding TL, Benet LZ (1979) Determination of 6-mercaptopurine and azathioprine in plasma by high-performance liquid chromatography. J Chromatogr 163: 281–288
Elion GB, Hitchings GH (1975) Azathioprine. In: Eichler O, Farah A, Herken H, Welch AD (eds) Handbook of experimental pharmacology, vol 38. Springer, New York
Erdmann GR, Chan GLC, Canafax DM (1988) HPLC determination of 6-thiouric acid and 6-mercaptopurine in organ transplant patient serum. J Liq Chromato 11: 971–981
Frey BM, Frey FJ, Benet LZ, Cochrum KC (1980) Modification of the mixed lymphocyte reaction for pharmacokinetic assessment of immunosuppressive activity in human plasma. Int J Immunopharmacol 2: 129–134
Haesslein HC, Pierce JC, Lee HM, Hume DM (1972) Leukopenia and azathioprine management in renal homotransplantation. Surgery 71: 598–604
Jackson PJ (1983) Determination of 6-thiouric acid in human urine. Clin Biochem 16: 285–286
Lavi LE, Holcenberg JS (1985) A rapid and sensitive high-performance liquid chromatographic assay for 6-mercaptopurine metabolites in red blood cells. Anal Biochem 144: 514–521
Lennard L, Keen D, Lilleyman JS (1986) Oral 6-mercaptopurine in childhood leukemia: Parent drug pharmacokinetics and active metabolite concentrations. Clin Pharmacol Ther 40: 287–292
Lennard L, Maddocks JL (1983) Assay of 6-mercaptopurine and 6-thioguanine in human red blood cells. J Pharm Pharmacol 35: 15–18
Lin S-N, Jessup K, Floyd M, Wang TPF, Van Buren CT, Caprioli RM, Kahan BD (1980) Quantitation of plasma azathioprine and 6-mercaptopurine levels in renal transplant patients. Transplantation 29: 290–294
Murray JE, Merrill JP, Harrison H, Wilson RE, Dammin GJ (1963) Prolonged survival of human-kidney homografts by immunosuppression drug therapy. N Engl J Med 268: 1315–1323
Odlind B, Hartvig P, Lindstrom B, Lonnerhohm G, Tufveson G, Grefgerg N (1986) Serum azathioprine and 6-mercaptopurine levels and immunosuppressive activity after azathioprine in uremic patients. Int J Immunopharmacol 8: 1–11
Pollack R, Nishikawa RA, Mozes MF, Jonasson O (1980) Azathioprine-induced leukopenia-clinical significance in renal transplantation. J Surg Res 29: 258–264
Simmons RL, Canafax DM, Fryd DS, Ascher NL, Payne WD, Sutherland DER, Najarian JS (1986) New immunosuppressive drug combinations for mismatched related and cadaveric renal transplantation. Transplant Proc 18 [Suppl 1]: 76–81
Sulh H, Koren G, Whalen C, Soldin S, Zipursky A, Greenberg M (1986) Pharmacokinetic determinants of 6-mercaptopurine myelotoxicity and therapeutic failure in children with acute lymphoblastic leukemia. Clin Pharmacol Ther 40: 604–609
Van Scoik KG, Johnson CA, Porter WR (1985) The pharmacology and metabolism of the thiopurine drugs 6-mercaptopurine and azathioprine. Drug Metab Rev 16: 157–174
Van Scoik KG, Johnson CA, Porter WR (1987) Sensitive and specific high-performance liquid chromatographic assay for 6-thiouric acid. J Chromatogr 417: 183–189
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chan, G.L.C., Erdmann, G.R., Gruber, S.A. et al. Pharmacokinetics of 6-thiouric acid and 6-mercaptopurine in renal allograft recipients after oral administration of azathioprine. Eur J Clin Pharmacol 36, 265–271 (1989). https://doi.org/10.1007/BF00558158
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00558158