Summary
Nine patients (6 males, 3 females) with biopsy-proven liver cirrhosis participated in an open, cross-over, three centre study of the effect of impaired liver function on the pharmacokinetics of felodipine. Two of the nine patients had undergone porto-caval anastomosis. Each patient was given 0.75 mg i.v. and 10 mg p.o. on separate occasions. The results of this study have been compared with published data from younger subjects and elderly hypertensive patients.
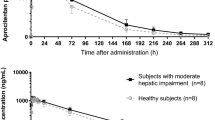
The mean peak plasma concentration normalized to a dose of 10 mg (Cmax 46 nmol/l) was twice as high in the cirrhotic patients as in the healthy subjects, but the bioavailability, f, (17.0%) was comparable. Subjects with a porto-caval shunt did not have higher f than the mean for the group. The volume of distribution at steady-state, Vss, was significantly lower than in the healthy subjects. Protein binding was significantly lower in the patients with cirrhosis: 99.46% compared to 99.64% in the healthy subjects. The weight-corrected clearance was 1/3 of the value in healthy subjects.
No correlation between systemic availability and oral clearance was found, so it is proposed that felodipine is metabolized both in the liver and also in the gut wall. The results suggest that at least the starting dose should be reduced in patients with severe liver disease.
Similar content being viewed by others
References
Ljung B (1985) Vascular selectivity of felodipine. Drugs 29 [Suppl 2]: 46–58
Nordlander M (1985) Haemodynamic effects of short and long term administration of felodipine in spontaneously hypertensive rats. Drugs 29 [Suppl 2]: 90–101
Edgar B, Bengtsson B, Elmfeldt D, Lundborg P, Nyberg G, Raner S, Rönn O (1985) Acute diuretic/natriuretic properties of felodipine in man. Drugs 29 [Suppl 2]: 176–184
Katzman PL, Hulthen UL, Hökfelt D (1986) The effect of 8 weeks treatment with the calcium antagonist felodipine on blood pressure, heart rate, working capacity, plasma renin activity, plasma angiotensin II, urinary catecholamines and aldosterone in patients with essential hypertension. Br J Clin Pharmacol 21: 633–640
Elmfeldt D, Hedner T (1983) Felodipine — A new vasodilator — in addition to β-receptor blockade in hypertension. Eur J Clin Pharmacol 25: 571–575
Co-operative Study Group (1986) Felodipine vs hydralazine: A controlled trial as third line therapy in hypertension. Br J Clin Pharmacol 21: 621–626
Edgar B, Regårdh CG, Johnsson G, Johansson L, Lundborg P, Löfberg I, Rönn O (1985) Felodipine kinetics in healthy man. Clin Pharmacol Ther 38: 205–211
Edgar B, Regårdh CG, Lundborg P, Romare S, Nyberg G, Rönn O (1987) Pharmacokinetic and pharmacodynamic studies of felodipine in healthy subjects after various single oral and intravenous doses. Biopharm Drug Dispos 8: 235–248
Edgar B, Collste P, Haglund K, Regårdh CG (1987) Pharmacokinetics and haemodynamic effect of felodipine as monotherapy in hypertensive patients. Clin Inv Med 10: 388–394
Landahl S, Edgar B, Gabrielsson M, Larsson M, Lernfelt B, Lundborg P, Regårdh CG (1988) Pharmacokinetics and blood pressure effect of felodipine in elderly hypertensive patients — a comparison with young healthy subjects. Clin Pharmacokinet 14: 374–383
Bäärnhielm C, Skånberg I, Borg KO (1984) Cytochrome P-450-dependent oxidation of felodipine — a 1.4-dihydropyridine to the corresponding pyridine. Xenobiotica 14: 719–726
Triggle DJ (1982) Biochemical pharmacology of calcium blockers. In: Flaim SF, Zelis R (eds) Calcium blockers: Mechanism of action and clinical application. Urban and Schwarzenberg, Baltimore-Munich, p 121
Ahnoff M, Ervik M, Johansson L (1987) Comparison of high-selectivity gas chromatographic methods, including column switching, for the determination of felodipine in plasma. Chromatography 394: 419–427
Peck CG, Beal SL, Sheiner LB (1984) Extended least squares nonlinear regression: A possible solution to the “choice of weights” problem in analysis of individual pharmacokinetic data. J Pharmacokinet Biopharm 12: 545–558
Sheiner LB, Beal SL (1985) Pharmacokinetic parameter estimates from seven least squares procedures: Superiority of extended least squares. J Pharmacokinet Biopharm 13: 185–201
Loo JCK, Riegelman S (1970) Assessments of pharmacokinetic constants from post infusion blood curves obtained after i.v. infusion. J Pharm Sci 59: 53–57
Edgar B, Regårdh CG, Attman PO, Aurell M, Herlitz H, Johnsson G (1989) Pharmacokinetics of felodipine in patients with impaired renal function. Br J Clin Pharmacol 27: 67–74
Jackson FC, Perrin EB, Felix WR, Smith AG (1971) The clinical investigation of the portocaval shunt. Survival analysis of the therapeutic operation. Am Surg 174: 672–698
Somogyi A, Eichelbaum M, Gugler R (1982) Prediction of bioavailability for drugs with high first-pass effect using oral clearance data. Eur J Clin Pharmacol 22: 85–90
Waller DG, Renwick AG, Gruchi BC (1984) The first-pass metabolism of nifedipine in man. Br J Clin Pharmacol 18: 951–954
Child CG, Turcotte JG (1964) Surgery and portal hypertension. In: Child CG (ed) The liver and portal hypertension. WB Saunders, Philadelphia, p 50
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Regårdh, C.G., Edgar, B., Olsson, R. et al. Pharmacokinetics of felodipine in patients with liver disease. Eur J Clin Pharmacol 36, 473–479 (1989). https://doi.org/10.1007/BF00558072
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00558072