Summary
We have studied the pharmacokinetics and haemodynamic effects of nicorandil after a 12-h infusion. Nicorandil is a mixed vasodilator combining the actions of a nitrate and a potassium channel opener.
Nicorandil was infused for 12 h in 21 healthy volunteers at rates of 0.05, 0.10, and 0.20 μg·kg−1·min−1 using a placebo controlled, crossover design. Systemic blood pressure, heart rate, electrocardiographic (ECG) intervals, and cardiac output (impedance cardiography) were measured supine and standing.
Dose-related, steady-state plasma nicorandil concentrations occurred within 3 to 4 h. Nicorandil's pharmacokinetics were linear with dose.
Four 0.20 μg·kg−1·min−1 nicorandil infusions were terminated early primarily because of moderate or severe headaches. There were no safety concerns (ECG intervals, laboratory assays).
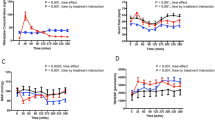
Blood pressure fell versus placebo only in the standing position and heart rate increased slightly (not significant). That is, standing blood pressure in the 6 to 12 h interval fell from baseline 8.0*/6.8, 1.6/5.1, and 9.8*/7.9* mmHg (systolic/diastolic, *=P<0.05 versus placebo) at 0.05, 0.10, and 0.20 μg·kg−1·min−1 respectively. Cardiac output increased slightly above placebo at lower doses. Haemodynamic changes correlated poorly with plasma nicorandil concentrations. Similar total doses were less well-tolerated when extended over 12 h. We saw no evidence of pharmacodynamic tolerance to nicorandil within 12 h.
Similar content being viewed by others
References
Sakai K, Nakano H, Nagano H, Uchida Y (1983) Nicorandil. In: Scriabine A (ed) New drugs annual: cardiovascular drugs. Raven Press, New York, pp 227–242
Kinoshita M, Sakai K (1990) Pharmacology and therapeutic effects of nicorandil. Cardiovasc Drugs Ther 4: 1075–1088
Belz GG, Matthews JH, Beck A, Wagner G, Schneider B (1985) Hemodynamic effects of nicorandil, isosorbide dinitrate, and dihydralazine in healthy volunteers. J Cardiovasc Pharmacol 7: 1107–1112
Belz GG, Matthews J, Heinrich J, Wagner G (1984) Controlled comparison of the pharmacodynamic effects of nicorandil (SG-75) and isosorbide dinitrate in man. Eur J Clin Pharmacol 26: 681–685
Coltart DJ, Signy M (1989) Acute hemodynamic effects of single-dose nicorandil in coronary artery disease. Am J Cardiol 63: 34J-39J
Solal AC, Jaeger P, Bouthier J, Juliard J-M, Dahan M, Gourgon R (1989) Hemodynamic action of nicorandil in chronic congestive heart failure. Am J Cardiol 63: 44J-48J
Tice FD, Binkley PF, Cody RJ, Moeschberger ML, Mohrland JS, Wolf DL, Leier CV (1990) Hemodynamic effects of oral nicorandil in congestive heart failure. Am J Cardiol 65: 1361–1367
Thormann J, Schlepper M, Kramer W, Gottwik M, Kindler M (1983) Effectiveness of nicorandil (SG-75), a new long-acting drug with nitroglycerin effects, in patients with coronary artery disease: improved left ventricular function and regional wall motion and abolition of pacing-induced angina. J Cardiovasc Pharmacol 5: 371–377
Frydman AM, Chapelle P, Diekmann H, Bruno R, Thebault JJ, Bouthier J, Caplain H, Ungethuem W, Gaillard C, Le Liboux A, Renard A, Gaillot J (1989) Pharmacokinetics of nicorandil. Am J Cardiol 63: 25J-33J
Wolf DL, Ferry JJ, Hearron AE, Froeschke MO, Luderer JR (1993) The haemodynamic effects and pharmacokinetics of intravenous nicorandil in healthy volunteers. Eur J Clin Pharmacol 44: 27–33
Venitz J, Lucker PW (1984) Impedance cardiography — a reliable method for measuring cardiac function noninvasively. Methods Find Exp Clin Pharmacol 6: 339–346
Searle SR (1987) Linear models for unbalanced data. Wiley, New York, pp 340–359
Snedecor GW, Cochran WG (1980) Statistical methods, 7th edn. The Iowa State University Press, Ames, pp 89–95, pp 175–178
Gibaldi M, Perrier D (1982) Pharmacokinetics, 2nd edn. Dekker, New York, pp 409–417
Imhof PR, Sieber A, Hodler J, Müller P, Ott B, Fankhauser P, Chu L-C, Gérardin A (1982) Plasma concentrations and haemodynamic effects of nitroglycerin during and after intravenous infusion in healthy volunteers. Eur J Clin Pharmacol 23: 99–106
Goldberg MR (1988) Clinical pharmacology of pinacidil, a prototype for drugs that affect potassium channels. J Cardiovasc Pharmacol 12 [Suppl 2]: S41-S47
Frydman A (1992) Pharmacokinetic profile of nicorandil in humans: an overview. J Cardiovasc Pharmacol 20 [Suppl 3]: S34-S44
Wilkinson GR (1975) Pharmacokinetics of drug disposition: hemodynamic considerations. Ann Rev Pharmacol 15: 11–27
Modi MW, Hassett JM, Lalka D (1988) Influence of posture on hepatic perfusion and the presystemic biotransformation of propranolol: simulation of the food effect. Clin Pharmacol Ther 44: 268–274
Schaumann W (1989) Pharmacokinetics of isosorbide dinitrate and isosorbide-5-mononitrate. Int J Clin Pharmacol Ther Toxicol 27: 445–453
Parker JD, Farrell B, Fenton T, Cohanim M, Parker JO (1991) Counter-regulatory responses to continuous and intermittent therapy with nitroglycerin. Circulation 84: 2336–2345
Belz GG, Beermann C (1992) Venodilatory effects of nicorandil in healthy volunteers. J Cardiovasc Pharmacol 20 [Suppl 3]: S57-S58
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wolf, D.L., Hearron, A.E., Metzler, C.M. et al. The pharmacokinetics and haemodynamic effects of continuous nicorandil infusion in healthy volunteers. Eur J Clin Pharmacol 45, 437–443 (1993). https://doi.org/10.1007/BF00315515
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00315515