Abstract
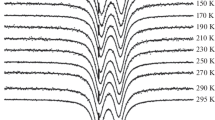
The mixed valence iron silicate deerite, Fe 2+6 Fe 3+3 O3[Si6O17](OH)5 whose crystal structure has only recently been fully determined, had been previously examined by both susceptibility measurements and Mössbauer spectroscopy. We reinterpret the Mössbauer measurements from Frank and Bunbury and show that all contradictions between the phenomena, observed in their experiments, and the mineralogical structure disappear when one accepts a thermally activated electron delocalisation associated with Fe2+ ↔ Fe3+ charge transfer.
Similar content being viewed by others
References
Asborne MD, Parkin KM, Burns RG (1978) Temperature dependence of Fe-Ti spectra in the visible region: Implication to mapping Ti concentrations of hot planetary surfaces Proc 9th Lunar Sci Conf 2949
Agrell SO, Bown MG, Mc Kie D (1965) Deerite, howiete, and zussmanite, three new minerals from the Francis and Laytonville District. Mendocino Co., California. Am Mineral 50:278
Bancroft GM (1973) Mössbauer spectroscopy, an introduction for inorganic chemists and geochemists. McGraw Hill, London, p 157
Bancroft GM, Burns RG, Stone AJ (1968) Application of the Mössbauer effect to silicate mineralogy — II, Iron silicates of unknown and complex crystal structures. Geochim Cosmochim Acta 32:547
Bunbury DStP, Frank E (1969) Mössbauer effect in deerite, Proc 11th Conf on coordination chemistry, September 1968. In: Cals M (ed) Progress in coordination chemistry. Elsevier, Amsterdam
Blume M, Tjon JA (1968) Mössbauer spectra in a fluctuating environment. Phys Rev 165:44
Carmichael IS, Fyfe WS, Machin DJ (1966) Low spin ferrous iron in the iron silicate deerite. Nature 221:1389
Evans BJ (1977) Melonjosephite, post-deadline oral presentation. Int Conf Appl Mössbauer Spectroscopy, Bucarest
Fleet ME (1977) The crystal structure of deerite. Am Mineral 62:990
Frank E, Bunbury DStP (1974) A study of deerite by the Mössbauer effect. J Inorg Nucl Chem 36:1725
Gérard A, Grandjean F (1971) Observation by Mössbauer effect of an electron hopping process in ilvaite. Solid State Commun 9:1845
Grandjean F, Gérard A (1975) Mössbauer spectra in the presence of a fluctuating electric field gradient. Application to the case of semimetallic compounds. Solid State Commun 16:553
Hartmann-Boutron F (1968) Effets des fluctuations du couplage quadrupole sur les spectres Mössbauer des ions dans les cristaux. J Phys 29:47
Jeffersen JH, Stevens KWH (1978) Intermediate valence — a view of the theoritical situation. J Phys C Solid State Phys 11:3919
Langer K, Lattard D, Schreyer W (1977) Synthesis and stability of deerite, Fe 2+12 Fe 3+6 [Si12O40] (OH)10, and Fe3+ ⇌ Al3+ substitution at 1528 kb. Contrib Mineral Petrol 60:271
Lotgering FK, Diepen AM van (1977) Electron exchange between Fe2+ and Fe3+ ions in octahedral sites in spinels studied by means of paramagnetic Mössbauer spectra and susceptibility measurements. J Phys Chem Solids 38:565
Nolet DA, Burns RG (1978) Temperature dependent Fe2+ ↔ Fe3+ electron delocalisation in ilvaite. Geophys Res Lett 5:821
Nolet DA, Burns GR (1979) Ilvaïte: A Study of temperature dependent electron delocalization by the Mössbauer effect. Phys Chem Mineral 4:221
Parkin KM, Loeffler BM, Burns RG (1977) Mössbauer spectra of kyanite, aquamarine, and cordierite, showing intervalence charge transfer. Phys Chem Mineral 1:301
Pollak H (1976) Charge transfer in cordierite. Phys Status Solidi B: 74:31
Shannon RD, Prewitt CT (1969) Effective ionic radii in oxides and fluorides. Acta Crystallogr Sect B: 25:925
Smith G, Strens RGJ (1976) Intervalence — transfer absorption in some silicate, oxide, and phosphate minerals. In: Strens RGJ (ed) The physics and chemistry of minerals and rocks. J. Wiley and Sons, London, pp 583–612
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pollak, H., Quartier, R. & Bruyneel, W. Electron relaxation in deerite. Phys Chem Minerals 7, 10–14 (1981). https://doi.org/10.1007/BF00308194
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00308194