Abstract
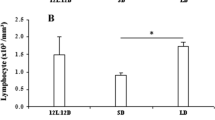
The effects of photoperiod and melatonin treatment on reproductive and immune function were assessed in two subspecies of Peromyscus maniculatus from different latitudes of origin. In experiment 1, P. m. bairidii (latitude = 42°51′ N) and P. m. luteus (latitude = 30°37′ N) were housed in either long (LD 16∶8) or short days (LD 8∶16) for 8 weeks. Short-day P. m. bairdii displayed reproductive regression and elevated splenocyte proliferation in response to the T-cell mitogen concanavalin A, as compared to long-day mice. In contrast, P. m. luteus did not undergo reproductive regression or exhibit any increase in lymphocyte proliferation in short days. In experiment 2, individuals of both P. m. bairdii and P. m. luteus were implanted with empty capsules or capsules that contained melatonin. Individual P. m. bairdii implanted with melatonin underwent reproductive regression. Individuals of this subspecies also displayed elevated lymphocyte proliferation compared to control mice. Conversely, P. m. luteus implanted with melatonin did not undergo reproductive regression and displayed no significant changes in lymphocyte proliferation. These results suggest that reproductive responsiveness to melatonin mediates short-day enhancement of immune function in deer mice. These data also imply that melatonin may not possess universal immunoenhancing properties. Rather, the effectiveness of melatonin to influence immune responses may be constrained by reproductive responsiveness to this indole-amine.
Similar content being viewed by others
References
Afoke AO, Eeg-Olofsson O, Hed J, Kjellman NM, Lindblom B, Ludvigsson J (1993) Seasonal variation and sex differences of circulating macrophages, immunoglobulins, and lymphocytes in healthy school children. Scand J Immunol 37: 209–215
Bartness TJ, Goldman BD (1989) Mammalian pineal melatonin: a clock for all seasons. Experientia 45: 939–945
Bartness TJ, Powers JB, Hastings MH, Bittman EL, Goldman BD (1993) The timed infusion paradigm for melatonin delivery: what has it taught us about the melatonin signal, its reception, and the photoperiodic control of seasonal responses? J Pineal Res 15: 161–190
Bittman EL, Dempsey RJ, Karsch FJ (1983) Pineal melatonin secretion drives the reproductive response to daylength in the ewe. Endocrinology 113: 2276–2283
Blank JL (1992) Phenotypic variation in physiological response to seasonal environments. In: Tomasi TE and Horton TH (eds) Mammalian energetics: interdisciplinary views of metabolism and reproduction. Cornell University Press, New York, pp 186–212
Blask DE, Lemus-Wilson ST, Cos S (1992) Neurohormonal modulation of cancer growth by pineal melatonin. In: Touitou Y et al. (eds) Melatonin and the pineal gland: from basic science to clinical application. Elsevier, Amsterdam, pp 303–310
Blom JMC, Gerber JM, Nelson RJ (1994) Day length affects immune function in deer mice: interactions with age, sex, and prenatal photoperiod. Am J Physiol 267: 596–601
Boctor FN, Charmy RA, Cooper EL (1989) Seasonal differences in the rhythmicity of human male and female lymphocyte blastogenic responses. Immunol Invest 18: 775–784
Brainard GC, Watson-Whitmyer M, Knobler RL, Lubin FD (1988) Neuroendocrine regulation of immune parameters: photoperiod control of the spleen in Syrian hamsters. Ann NY Acad Sci 540: 704–706
Bronson FH (1989) Mammalian reproductive biology. University of Chicago Press, New York
Bronson FH, Heideman PD (1994) Seasonal regulation of reproduction in mammals. In: Knobil E, Neill JD (eds) The physiology of reproduction, vol 2, 2nd edn. Raven Press, New York, pp 541–584
Calvo JR, Rafii-El-Idrissi M, Pozo D, Guerrero JM (1995) Immunomodulatory role of melatonin: specific binding sites in human and rodent lymphoid cells. J Pineal Res 18: 119–126
Carlson LL, Zimmerman A, Lynch GR (1989) Geographic differences for delay of sexual maturation in Peromyscus leucopus: effects of photoperiod, pinealectomy, and melatonin. Biol Reprod 41: 1004–1013
Carter DS, Goldman BD (1983) Antigonadal effects of timed melatonin infusion in pinealectomized male Djungarian hamsters (Phodopus sungorus sungorus): duration is the critical parameter. Endocrinology 113: 1261–1267
Cory AH, Owen TC, Barltrop JA, Cory JG (1991) Use of an aqueous soluble tetrazolium/formazan assay for cell growth assays in culture. Cancer Commun 3: 207–212
Denias GE, Nelson RJ (1996) Photoperiod and temperature interact to affect immune parameters in adult deer mice (Peromyscus maniculatus). J Biol Rhythms 11: 94–102
Desjardins C, Lopez MJ (1980) In: Steinberger A, Steinberger E (eds) Sensory and nonsensory modulation of testis function. In: Steinberger A, Steinberger E (eds) Testicular Development, Structure and Function. Raven Press, New York, pp 381–388
Fairbairn DJ (1977) Why breed early? A study of reproductive tactics in Peromyscus. Can J Zool 55: 862–871
Goldman BD, Darrow JM (1983) The pineal gland and mammalian photoperiodism. Neuroendocrinology 37: 386–396
Goldman BD, Nelson RJ (1983) Melatonin and seasonality in mammals. In: Yu HS, Reiter RJ (eds) Melatonin: biosynthesis, physiological effects and clinical applications. CRC Press, New York, pp 225–252
Guerrero JM, Reiter RJ (1992) A brief survey of pineal glandimmune system interrelationships. Endocrinol Res 18: 91–113
Heath HW, Lynch GR (1982) Intraspecific differences for melatonin-induced reproductive regression and the seasonal molt in Peromyscus leucopus. Gen Comp Endocrinol 48: 289–295
Heldmaier G, Steinlechner S, Ruf T, Wiesinger H, Klingenspor M (1989) Photoperiod and thermoregulation in vertebrates: body temperature rhythms and thermogenic acclimation. J Biol Rhythms 4: 351–365
John JL (1994) The avian spleen: a neglected organ. Q Rev Biol 69: 327–351
Lochmiller RL, Vestey MR, McMurray ST (1994) Temporal variation in humoral and cell-mediated immune response in a Sigmodon hispidus population. Ecology 75: 236–245
Lopez-Gonzales MA, Calvo JR, Osuna C, Guerrero JM (1992) Interaction of melatonin with human lymphocytes: evidence for binding sites coupled to potentiation of cyclic AMP stimulated vasoactive intestinal peptide and activation of cyclic GMP. J Pineal Res 12: 97–104
Lynch GR, Epstein AL (1976) Melatonin-induced changes in gonads, pelage and thermogenic characters in the white-footed mouse Peromyscus leucopus. Comp Biochem Physiol C 53: 67–68
Lynch GR, Sullivan JK, Gendler SL (1980) Temperature regulation in the mouse, Peromyscus leucopus: effects of various photoperiods, pinealectomy, and melatonin administration. Int J Biometeorol 24: 49–55
Lynch GR, Heath HM, Johnston CM (1981) Effect of geographic origin on the photoperiodic control of reproduction in the white-footed mouse, Peromyscus leucopus. Biol Reprod 25: 475–180
Masestroni GJ (1993) The immunoendocrine role of melatonin. J Pineal Res 14: 1–10
Moffatt CA, De Vries AC, Nelson RJ (1993) Winter adaptations of male deer mice (Peromyscus maniculatus) and prairie voles (Microtus ochrogaster) that vary in reproductive responsiveness to photoperiod. J Biol Rhythms 8: 221–232
Nelson RJ, Badura LL, Goldman BD (1990) Mechanisms of seasonal cycles of behavior. Annu Rev Psychol 41: 81–109
Nelson RJ, Blom JMC (1994) Photoperiodic effects on tumor development and immune function. J Biol Rhythms 9: 233–249
Nelson RJ, Demas GE (1996) Seasonal changes in immune function. Q Rev Biol (in press).
Nelson RJ, Demas GE, Klein SL, Kriegsfeld LJ (1995) The influence of season, photoperiod, and pineal melatonin on immune function. J Pineal Res 19: 149–165
Petterborg LJ, Reiter RJ (1981) Effects of photoperiod and subcutaneous melatonin implants on the reproductive status of adult white-footed mice (Peromyscus leucopus). J Androl 2: 222–224
Rafii-El-Idrissi M, Calvo JR, Pozo D, Harmouch A, Guerrero JM (1995) Specific binding of 2-[125I] iodomelatonin by rat splenocytes: characterization and its role on regulation of cyclic AMP production. J Neuroimmunol 57: 171–178
Sullivan JK, Lynch GR (1986) Photoperiod time measurement for activity, torpor, molt and reproduction in mice. Physiol Behav 36: 167–174
Tamarkin L, Cohen M, Roselle D, Reichert C, Lippman M, Chabner B (1981) Melatonin inhibition and pineal enhancement of 7,12-dimethyl-benzanthracene-induced mammary tumors in the rat. Cancer Res 41: 4432–4436
Vaughan MK, Hubbard GB, Champney TH, Vaughan GM, Little JC (1987) Splenic hypertrophy and extramedullary hematopoiesis induced in male Syrian hamsters by short photoperiod or melatonin injections and reversed by melatonin pellets or pinealectomy. Am J Anat 179: 131–136
Vriend J, Lauber JK (1973) Effects of light intensity, wavelength and quanta on gonads and spleen of the deer mouse. Nature 244: 37–38
Weaver DR, Provencio I, Carlson LL, Reppert SM (1991) Melatonin receptors and signal transduction in photorefractory Siberian hamsters (Phodopus sungorus). Endocrinology 128: 1086–1092
Weaver DR, Rivkees SA, Reppert SM (1989) Localization and characterization of melatonin receptors in rodent brain by in vitro autoradiography. J Neurosci 9: 2581–2590
Wunder BA (1992) Morphophysiological indicators of the energy state of small mammals. In: Tomasi TE, Horton TH (eds) Mammalian energetics: interdisciplinary views of metabolism and reproduction. Cornell University Press, New York, pp 86–104
Wurtman RJ, Weisel J (1969) Environmental lighting and neuroendocrine function: relationship between spectrum of light source and gonadal growth. Endocrinology 85: 1218–1221
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Demas, G.E., Klein, S.L. & Nelson, R.J. Reproductive and immune responses to photoperiod and melatonin are linked in Peromyscus subspecies. J Comp Physiol A 179, 819–825 (1996). https://doi.org/10.1007/BF00207360
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00207360