Summary
5-Fluorouracil (FU) and its masked compounds tegafur (FT) and carmofur (HCFU) were administered orally to Beagle dogs daily for 6 months, and their chronic neurotoxic effects were examined morphologically.
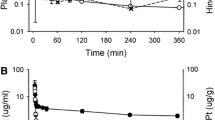
In ten dogs that survived the 6-month treatment large vacuoles produced by splitting of the intraperiod line of myelin were observed in the fornix in the wall of the third ventricle. In severely affected dogs large vacuoles developed in the medial preoptic area, medial portion of the internal capsule, the area around the subthalamic nucleus and the mammillo-thalamic tract. Axons of myelinated fibers affected by vacuolation were generally well maintained, and destruction of myelin was not detected. Though proliferation of glia cells or abnormality of oligodendroglia was not detected, a lipid deposit covered by a single layer membrane was observed in the cell bodies and processes of astrocytes. No abnormality was detected by electron microscopy in the cerebrum, inferior colliculus, cerebellum, or pons. Of eight dogs that died during the treatment period, large vacuoles were observed in the fornix in the wall of the third ventricle of four dogs treated for more than 1 month, and large vacuoles were present in the inferior colliculus in two dogs of the FT group in the above four dogs. In the HCFU group, the interruption of treatment for 6months resulted in alleviation or disappearance of the vacuolar lesions. The above findings suggest that the neurotoxicity of FU and its masked compounds FT and HCFU in long-term treatment produces changes morphologically identical with one another in respect to the site of their manifestation and nature of lesion, that their common degraded product α-fluoro-β-alanine (FBAL) plays a crucial role in their neurotoxic actions, and that vacuolar lesions, to which myelin was more vulnerable than neurons, can develop where the toxic substance readily deposits and accumulates.
Similar content being viewed by others
References
Bergevin PR, Patwardhan VC, Weissman J, Lee SM (1975) Neurotoxicity of 5-fluorouracil. Lancet I:410
Bixenman WW, Nicholls JVV, Warwick OH (1977) Oculomotor disturbances associated with 5-fluorouracil chemotherapy. Am J Ophthalmol 83:789–793
Boileau G, Piro AJ, Lahiri SR, Hall TC (1971) Cerebellar ataxia during 5-fluorouracil (NSC-19893) therapy. Cancer Chemother Rep 55:595–598
Bourke RS, Charles RW, Chheda G, Tower DB (1973) Kinetics of entry and distribution of 5-fluorouracil in cerebrospinal fluid and brain following intravenous injection in a primate. Cancer Res 33:1735–1746
Chadwick M, Rogers WI (1972) The physiological disposition of 5-fluorouracil in mice bearing solid L1210 lymphocytic leukemia. Cancer Res 32:1045–1056
Gottlieb JA, Luce JK (1971) Cerebellar ataxia with weekly 5-fluorrouracil administration. Lancet 1:138–139
Greco CM, Powell HC, Garrett RS, Lampert PW (1980) Cycloleucine encephalopathy. Neuropathol Appl Neurol 6:349–360
Greenwald ES (1976) Organic mental changes with fluorouracil therapy. JAMA 235:248–249
Henni Zen-ichi, Nishina T, Yoshihara K, Ishi M, Tanaka Y, Kitano T (1980) General pharmacological studies on 1-hexylcarbamoyl-5-fluorouracil (HCFU), a new antitumor agent. Oyo Yakuri 19:369–382
Hirano A, Dembitzer HM (1967) A structural analysis of the myelin sheath in the central nervous system. J Cell Biol 34:555–567
Hirano A, Zimmerman HM, Levine S (1968) Intramyelinic and extracellular spaces in thriethyltin intoxication. J Neuropathol Exp Neurol 27:571–580
Hirano A, Dembitzer HM, Zimmerman HM (1969) The distribution of peroxidase in the triethyltin-intoxicated rat brain. J Neuropathol Exp Neurol 28:507–511
Horton J, Olson KB, Sullivan J, Reilly C, Shnider B, The Eastern Cooperative Oncology Group (1970) 5-Fluorouracil in cancer: An improved regimen. Ann Int Med 73:897–900
Ishimura K, Toizumi S, Neda K, Inoue H, Sato H, Sato K, Niyokawa H (1979) toxicological study on 1-hexylcarbamoyl-5-fluorouracil (HCFU). 2. Subacute and chronic toxicity studies in dogs. Pharmacometrics 17:597–615
Kimbrough RD (1973) Review of recent evidence of toxic effects of hexachlorophene. Pediatrics 51:391–394
Kobari T, Tan K, Kumakura M, Watanabe S, Shirakawa I, Kobayashi H, Ujiie A, Miyama Y, Namekawa H, Yamamoto H (1978) Metabolic fate of 1-hexylcarbamoyl-5-fluorouracil in rats. Xenobiotical 8:547–556
Kobari T, Iguro Y, Ujiie A, Namekawa H (1981) Metabolism of 1-hexylcarbamoyl-5-fluorouracil (HCFU), a new antitumour agent, in rats, rabbits, and dogs. Xenobiotica 11:57–62
Koenig H (1969) Acute axonal dystrophy caused by fluorocitrate: The role of mitochondrial swelling. Science 164:310–312
Koenig H, Patel A (1969) Biochemical basis of the acute cerebellar syndrome in 5-fluorouracil chemotherapy. Trans Am Neurol Assoc 94:290–292
Koenig H, Pattel A (1970) Biochemical basis for fluorouracil neurotoxicity. The role of Krebs cycle inhibition by fluoroacetate. Arch Neurol 23:155–160
Lynch HT, Droszcz CP, Albano WA, Lynch JF (1981) “Organic brain syndrome” secondary to 5-fluorouracil toxicity. Dis Colon Rectum 24:130–131
MacDonell LA, Potter PE, Leslie RA (1978) Localized changes in blood-brain barrier permeability following the administration of antineoplastic drugs. Cancer Res 38:2930–2934
Moertel CG, Reitemeier RJ, Bolton CF, Shorter RG (1964) Cerebellar ataxia associated with fluorinated pyrimidine therapy. Cancer Chemother Rep 41:15–18
Mukherjee KL, Heidelberger C (1960) Studies on fluorinated pyrimidines. IX. The degradation of 5-fluorouracil-6-C14. J Biol Chem 235:433–437
Mukherjee KL, Boohar J, Wentland D, Ansfield FJ, Heidelberger C (1963) Studies on fluorinated pyrimidines. XVI. Metabolism of 5-fluorouracil-2-C14 and 5-fluoro-2′-deoxyuridine-2-C14 in cancer patients. Cancer Res 23:49–66
Mullick FG (1973) Hexachlorophene toxicity—human experience at the Armed Forces Institute of Pathology. Pediatrics 51:395–399
Oishi T, Shiraki H, Mineura K, Takahira H (1973) The study on metabolic fate of 5-fluorouracil-6-14C after percutaneous administration (in Japanese). Yakugaku Zasshi 93:749–755
Palay SL, Chan-Palay V (1974) Cerebellar cortex: cytology and organization. Springer, Berlin Heidelberg New York
Pattison FLM, Peters RA (1966) Monofluoro aliphatic compounds. Handbool of experimental pharmacology, vol 20, part 1. Springer, Berlin Heidelberg New York, pp 387–458
Riehl J-L, Brown WJ (1964) Acute cerebellar syndrome secondary to 5-fluorouracil therapy. Neurology 14:961–967
Scriver CR, Pueschel S, Davies E (1966) Hyper-β-alaninemia associated with β-aminoaciduria and γ-aminobutyric acid uria, somnolence and seizures. New Engl J Med 274:635–643
Sueyoshi K, Uozumi A, Majima H (1980) Concentration of 1-hexylcarbamoyl-5-fluorouracil (HCFU) fraction and 5-fluorouracil in central nervous system after HCFU oral administration (in Japanese). Gan to kagakuryoho 7:870–874
Tripier MF, Berard M, Toga M, Martin-Bouyer G, Le Breton R, Garat J (1981) Hexachlorophene and the central nervous system. Toxic effects in mice and baboons. Acta Neuropathol (Berl) 53:65–74
Towfighi J, Gonatas NK, McCree L (1973) Hexachlorophene neuropathy in rats. Lab Invest 29:428–436
Weiss HD, Walker MD, Wiernik PH (1974) Neurotoxicity of commonly used antineoplastic agents (first of two parts). New Engl J Med 291:75–81
Yabuuchi Y, Osame Y, Takikawa K, Fujii T, Ozaki T, Harima, K (1971) General pharmacological studies of N1-(2-tetrahydrofuryl)-5-fluorouracil, FT-207, a new anticancerous agent. (in Japanese with english summary Pharmacometrics 5:569–584
Yamashita K, Fukunaga Y, Maruden A, Takenouchi T, Morita K (1980) Subacute toxicity of UFT, a new anti-cancer agent orally administrated in dogs (in Japanese). Pharmacometrics 20:973–999
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Okeda, R., Karakama, T., Kimura, S. et al. Neuropathologic study on chronic neurotoxicity of 5-fluorouracil and its masked compounds in dogs. Acta Neuropathol 63, 334–343 (1984). https://doi.org/10.1007/BF00687342
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00687342