Summary
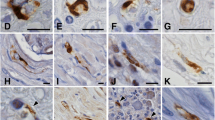
A novel type of non-viral cytoplasmic inclusion is described, which was seen in virtually every neuron in the brain and spinal cord of a child with a presumed metabolic disorder whose clinical picture and CNS pathology were compatible with Leigh Syndrome. The ovoid to round inclusions were sharply demarcated, measuring up to 11 μm in diameter. They showed no distinctive staining with a battery of routine histological techniques. The ultrastructural features are unique, comprising non-membrane-bounded aggregates of randomly oriented plate-like structures with parallel linear densities depicting a periodicity of 11–16 nm. Immunocytochemical studies revealed strong staining with antisera to tropomyosin and weaker staining with antisera to actin. There was no reactivity with antibodies against neurofilaments, microtubules and their associated proteins, paired helical filaments, ubiquitin, vinculin or alpha-actinin. It is postulated that the metabolic disorder resulted in a neurodegenerative condition which manifested pathologically with lesions compatible with those of Leigh Syndrome. Associated with the condition was the discrete accumulation of cytoplasmic proteinaceous components, including tropomyosin, in the form of neuronal cytoplasmic inclusions possibly resulting from an alteration of the neuronal cytoskeleton.
Similar content being viewed by others
References
Bloom GS, Luca FC, Vallee RB (1985) Microtubule-associated protein 1B: identification of a major component of the neuronal cytoskeleton. Proc Natl Acad Sci USA 82:5404–5408
Collins GH, Cowden RR, Nevis AH (1968) Myoclonus epilepsy with Lafora bodies. Arch Pathol 86:239–254
Culebras A, Feldman RG, Merk FB (1973) Cytoplasmic inclusion bodies within neurons of the thalamus in myotonic dystrophy. A light and electron microscope study. J Neurol Sci 19:319–329
David RB, Mamunes P, Rosenblum WI (1976) Necrotizing encephalomyelopathy (Leigh). In: Vinken PJ, Bruyn GW (eds) Handbook of clinical neurology, vol 28, chapter 14. North-Holland, Amsterdam, pp 349–363
De Camilli P, Miller PE, Navone F, Theurkauf WE, Vallee RB (1984) Distribution of microtubule-associated protein 2 in the nervous system of the rat studied by immunofluorescence. Neuroscience 11:819–846
Di Mauro S, Servidei S, Zeviani M, Di Rocco M, De Vivo DC, Di Donato S, Uziel G, Berry K, Hoganson G, Johnsen SD, Johnson PC (1987) Cytochromec oxidase deficiency in Leigh syndrome. Ann Neurol 22:498–506
Duffy PE, Tennyson VM (1965) Phase and electron microscopic observations of Lewy bodies and melanin granules in the substantia nigra and locus caeruleus in Parkinson's disease. J Neuropathol Exp Neurol 24:398–414
Galloway P, Perry G (1987) Microfilament involvement in Hirano body formation. In: Perry G (ed) Alterations in the neuronal cytoskeleton in Alzheimer disease. Plenum Press, New York, pp 199–210
Galloway PG, Perry G, Gambetti P (1987) Hirano body filaments contain actin and actin-associated proteins. J Neuropathol Exp Neurol 46:185–199
Galloway PG, Padget H, Perry G (1989) Alzheimer neurofibrillary tangles share epitopes with tropomyosin. (in preparation)
Gibson PH, Tomlinson BE (1977) Numbers of Hirano bodies in the hippocampus of normal and demented people with Alzheimer's disease. J Neurol Sci 33:199–206
Gilbert EF, Arya S, Chun R (1983) Leigh's necrotizing encephalopathy with pyruvate carboxylase deficiency. Arch Pathol Lab Med 107:162–166
Giometti CS, Anderson NL (1984) Tropomyosin heterogeneity in human cells. J Biol Chem 259:14113–14120
Goldman JE (1986) Cytoskeletal constituents of Hirano bodies. J Neuropathol Exp Neurol 45:334a
Hirano A, Malamud N, Elizan TS, Kurland LT (1966) Amyotrophic lateral sclerosis and Parkinsonism-dementia complex on Guam. Arch Neurol 15:35–51
Hirano A, Dembitzer HM, Kurland LT, Zimmerman HM (1968) The fine structure of some intraganglionic alterations. Neurofibrillary tangles, granulovascuolar bodies and “rod like” structures as seen in Guam amyotrophic lateral sclerosis and Parkinsonism-dementia complex. J Neuropathol Exp Neurol 27:167–182
Hitchcock-De Gregori SE, Heald RW (1987) Altered actin and troponin binding of amino-terminal variants of chicken striated muscle α-tropomyosin expressed inEscherichia coli. J Biol Chem 262:9730–9735
Hitchock-De Gregori SE, Lewis SF, Chou TM-T (1985) Tropomyosin lysine reactivities and relationship to coiledcoil structure. Biochemistry 24:3305–3314
Ihara Y, Abraham C, Selkoe DJ (1983) Antibodies to paired helical filaments in Alzheimer's disease do not recognize normal brain proteins. Nature 304:727–730
Joyce NC, Haire MF, Palade GE (1985) Contractile proteins in pericytes. I. Immunoperoxidase localization of the tropomyosin. J Cell Biol 100:1379–1386
Kosik KS, Duffy LK, Dowling MM, Abraham C, McCluskey A, Selkoe DJ (1984) Microtubule-associated protein 2: Monoclonal antibodies demonstrate the selective incorporation of certain epitopes into alzheimer neurofibrillary tangles. Proc Natl Acad Sci USA 81:7941–7945
Kosik KS, Joachim CL, Selkoe DJ (1986) Microtubule-associated protein tau (τ) is a major antigenic component of paired helical filaments in Alzheimer disease. Proc Natl Acad Sci USA 83:4044–4048
Manetto V, Perry G, Tabaton M, Mulvihill P, Fried VA, Smith HT, Gambetti P, Autilio-Gambetti L (1988) Ubiquitin is associated with abnormal cytoplasmic filaments characteristic of neurodegenerative diseases. Proc Natl Acad Sci USA 85:4501–4505
Matsumura F, Yamashiro-Matsumura S (1983) Isolation and characterization of tropomyosin-containing microfilaments from cultured cells. J Biol Chem 258:6636–6644
Montpetit VJA, Andermann F, Carpenter S, Fawcett JS, Aborowska-Sluis D, Giberson H (1971) Subacute necrotizing encephalomyelopathy — a review and a study of two families. Brain 94:1–30
Munoz DG, Greene C, Perl DP, Selkoe DJ (1988) Accumulation of phosphorylated neurofilaments in anterior horn motoneurons of amyotrophic lateral sclerosis patients. J Neuropathol Exp Neurol 47:9–18
Munoz-Garcia D, Pendlebury WW, Kessler JB, Perl DP (1986) An immunocytochemical comparison of cytoskeletal proteins in aluminium-induced and Alzheimer-type neurofibrillary tangles. Acta Neuropathol (Berl) 70:243–248
Onorato M, Conolly J, Kosik K, Perry G (1987) Altered neurites of Alzheimer disease contain a distinct subset of filaments sharing epitopes with tau. J Cell Biol 105:203a
Perry G, Manetto V, Onorato M, Weiss M, Mulvihill P, Galloway P, Tabaton M, Autilio-Gambetti L, Gambetti P (1987) Alteration of the neurofilament-microtubule network in Alzheimer disease and other neurodegenerative disorders. In: Perry G (ed) Alterations in the neuronal cytoskeleton in Alzheimer disease, Plenum Press, New York, pp 137–149
Peters A, Palay SL (1966) The morphology of laminae A and A1 of the dorsal nucleus of the lateral geniculate body of the cat. J Anat 100:451–486
Rezek PR, Millard M (1963) Autopsy pathology. A guide for pathologists and clinicians. Thomas, Springfield, p 780
Sawtell NW, Lessard JL, Hartman AL (1987) An immunologically defined marker of actin isoform divergence. J Cell Biol 105:26a
Schochet SS, Hardman JM, Ladewig PP, Earle KM (1969) Intraneuronal conglomerates in sporadic motor neuron disease. Arch Neurol 20:548–553
Schochet SS, Wyatt RB, McCormick F (1970) Intracytoplasmic acidophilic granules in the substantia nigra. A light and electron microscopic study. Arch Neurol 22:550–555
Stansbie D, Wallace SJ, Marsac C (1986) Disorders of the Pyruvate dehydrogenase complex. J Inherited Metab Dis 9:105–119
Sternberger LA (1986) Immunocytochemistry, 3rd edn. Wiley, New York
Stossel TP, Chaponnier C, Ezzell RM, Hartwig JH, Janmey PA, Kwiatkowski DJ, Lind SE, Smith DB, Southwick FS, Yin HL, Zaner KS (1985) Nonmuscle actin-binding proteins. Annu Rev Cell Biol 1:353–402
Takahashi K, Nakamura H, Okada E (1972) Hereditary amyotrophic lateral sclerosis. Histochemical and electron microscopic study of hyaline inclusions in motor neurons. Arch Neurol 27:292–299
Turel AP, Morris W, Levinsohn MW, Derakhshan I, Gutierrez Y (1975) Reye Syndrome and cerebellar intracytoplasmic inclusion bodies. Arch Neurol 32:624–628
Vallee RB, Dibartolomeis MJ, Theurkauf WE (1981) A protein kinase bound to the projection portion of MAP2 (microtubule-associated protein 2). J Cell Biol 90:568–576
White CI, Sparkeman DR, Gause JM (1985) Distribution of paired helical filament immunoreactivity in Alzheimertype dementia. J Neuropathol Exp Neurol 44:368
Wiśniewski HM, Berry K, Spiro AJ (1975) Ultrastructure of thalamic neuronal inclusions in myotonic dystrophy. J Neurol Sci 24:321–329
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lew, E.O., Rozdilsky, B., Munoz, D.G. et al. A new type of neuronal cytoplasmic inclusion: histological, ultrastructural, and immunocytochemical studies. Acta Neuropathol 77, 599–604 (1989). https://doi.org/10.1007/BF00687887
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00687887