Summary
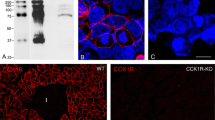
Using a specific polyclonal antibody raised against rat pancreatic phospholipase A2 (PLA2), we investigated the localization of the enzyme in the rat pancreas and stomach by light and electron microscopy. In the pancreas, the enzyme was localized in the acinar cells, whereas the pancreatic islets showed no immunoreaction. In the stomach, the PLA2 reactive with the anti-pancreatic PLA2 antibody was distributed exclusively in the gastric glands, but not in the gastric pits or the pyloric glands. On the section of the stomach subjected to immuno- and PAS-staining, immunopositive cells were not the PAS-positive cells located in the gastric pit and the neck region of the gastric gland.
Immunopositive cells were present from the neck to the bottom of the gastric gland. Immunoelectron microscopic observation revealed that the immunogold-labeled cell had a highly-developed rough endoplasmic reticulum in the basal cytoplasm and characteristic zymogen granules in the apical cytoplasm. Taking into account the cell position in the gastric gland, the immunopositive cell could therefore be identified as a chief cell. Since no double stainability with PLA2 and PAS was observed in the same cell, it is suggested that PLA2 could be used cytochemically as a marker enzyme of the chief cell in the gastric gland at the light-microscopic level. From the immunoelectron microscopic findings, we believe that the PLA2 in the stomach is released into the lumen of the stomach by exocytosis and could function as a digestive enzyme in the alimentary tract, like the PLA2 secreted from the pancreas. Other possible roles of the PLA2 in the stomach are discussed.
Similar content being viewed by others
References
Aase S, Dahl E, Roland M, Hars R (1983) Morphometric studies of parietal cells during basal conditions and during stimulation with pentagastrin in healthy subjects. Scand J Gastroenterol 18:913–923
Black JA, Forte TM, Forte JG (1980) Structure of oxyntic cell membranes during conditions of rest and secretion of HCl as revealed by freeze-fracture. Anat Rec 196:153–172
Forst S, Weiss J, Elsbach P (1986) Structural and functional properties of a phospholipase A2 purified from an inflammatory exudate. Biochemistry 25:8381–8385
Grataroli R, Charbonnier M, Leonardi J, Grimaud J, Lafont H, Nalbone G (1987) Phospholipase A2 activity of rat stomach. Arch Biochem Biophys 258:77–84
Hayakawa M, Horigome K, Kudo I, Tomita M, Nojima S, Inoue K (1987) Amino acid composition and NH2-terminal amino acid sequence of rat platelet secretory phospholipase A2. J Biochem 101:1311–1314
Helander HF, Leth R, Olbe L (1986). Stereological investigations on human gastric mucosa: I. Normal oxyntic mucosa. Anat Rec 216:373–380
Hirohara J (1987) Study of phospholipase in stomach: phospholipase A2 activity in rat stomach — its localization and effect of anti-ulcer and ulcerogenic agents on the activity. J Kansai Med Univ 39:119–136 (in Japanese)
Hirohara J, Sugatani J, Okumura T, Sameshima Y, Saito K (1987) Properties and localization of phospholipase A2 activity in rat stomach. Biochim Biophys Acta 919:231–238
Ishizaki J, Ohara O, Nakamura E, Tamaki M, Ono T, Kanda A, Yoshida N, Teraoka H, Tojo H, Okamoto M (1989) cDNA cloning and sequence determination of rat membrane-associated phospholipase A2 Biochem Biophys Res Commun 162:1030–1036
Kunze H (1970) Formation of [I-14C]prostaglandin E2 and two prostaglandin metabolites from [I-14C]arachidonic acid during vascular perfusion of the frog intestine. Biochim Biophys Acta 202:180–183
Lands WEM (1979) The biosynthesis and metabolism of prostaglandins. Annu Rev Physiol 41:633–652
Lands WEM, Samuelsson B (1968) Phospholipid precursors of prostaglandins. Biochim Biophys Acta 164:426–429
Matsuda Y, Ogawa M, Shibata T, Nakaguchi K, Nishijima J, Wakasugi C, Mori T (1987) Distribution of immunoreactive pancreatic phospholipase A2 (IPPL-2) in various human tissues. Res commun Chem Pathol Pharmacol 58:281–284
Ohara O, Tamaki M, Nakamura E, Tsuruta Y, Fujii Y, Shin M, Teraoka H, Okamoto M (1986) Dog and rat pancreatic phospholipases A2: complete amino acid sequences deduced from complementary DNAs. J Biochem 99:733–739
Okamoto M, Ono T, Tojo H, Yamano T (1985) Immunochemical relatedness between secretory phospholipase A2 and intracellular phospholipase A2. Biochem Biophys Res Commun 2:788–794
Ono T, Tojo H, Inoue K, Kagamiyama H, Yamano T, Okamoto M (1984) Rat pancreatic phospholipase A2: purification, characterization, andN-terminal amino acid sequence. J Biochem 96:785–792
Ono T, Tojo H, Kuramitsu S, Kagamiyama H, Okamoto M (1988) Purification and characterization of a membrane-associated phospholipase A2 from rat spleen: its comparison with a cytosolic phospholipase A2 S-1. J Biol Chem 263:5732–5738
Perlman D, Halvorson HO (1983) A putative signal peptidase recognition site and sequence in eukaryotic and prokaryotic signal peptides. J Mol Biol 167: 391–409
Postius S, Ruoff HJ, Szelenyi I (1985) Prostaglandin formation by isolated gastric parietal and nonparietal cells of the rat. Br J Pharmacol 84:871–877
Robert A (1979) Cytoprotection by prostaglandins. Gastroenterology 77:761–767
Robert A, Nezamis JE, Phillips JP (1967) Inhibition of gastric secretion by prostaglandins. Am J Dig Dis 12:1073–1076
Robert A, Nezamis JE, Phillips JP (1968) Effect of prostaglandin E1 on gastric secretion and ulcer formation in the rat. Gastroenterology 55:481–487
Robert A, Nylander B, Andersson S (1974) Marked inhibition of gastric secretion by two prostaglandin analogs given orally to man. Life Sci 14:533–538
Robert A, Schultz JR, Nezamis JE, Lancaster C (1976) Gastric antisecretory and antiulcer properties of PGE2, 15-methyl PGE2 and 16,16-dimethyl PGE2. Intravenous, oral and intrajejunal administration. Gastroenterology 70:359–370
Robert A, Nezamis JE, Lancaster C, Hanchar AJ (1979) Cytoprotection by prostaglandins in rats: prevention of gastric necrosis produced by alcohol, HCl, NaOH, hypertonic NaCl, and thermal injury. Gastroenterology 77:433–443
Sakata T, Nakamura E, Tsuruta Y, Tamaki M, Teraoka H, Tojo H, Ono T, Okamoto M (1989) Presence of pancreatic-type phospholipase A2 mRNA in rat gastric mucosa and lung. Biochim Biophys Acta 1007:124–126
Smolka A, Helander HF, Sachs G (1983) Monoclonal antibodies against gastric H+, K+-ATPase. Am J Physiol 245:G589-G596
Suzuki S, Furihata C, Tsuyama S, Murata F (1983a) Immunocyto-chemical localization of pepsinogen in rat stomach. Histochemistry 79:167–176
Suzuki S, Tsuyama S, Murata F (1983b) Cells intermediate between mocous neck cells and chief cells in rat stomach. Cell Tissue Res 233:475–484
Tarnawski A, Ivey KJ, Krause WJ, Sherman D, Burks M, Hewett J (1980) Quantitative analysis of human parietal cells after pentagastrin: correlation with gastric potential difference. Lab Invest 42:420–426
Tojo H, Ono T, Kuramitsu S, Kagamiyama H, Okamoto M (1988a) A phospholipase A2 in the supernatnat fraction of rat spleen: its similarity to rat pancreatic phospholipase A2. J Biol Chem 263:5724–5731
Tojo H, Ono T, Okamoto M (1988b) A pancreatic-type phospholipase A2 in rat gastric mucosa. Biochem Biophys Res Commun 151:1188–1193
Vadas P, Prauzanski W (1986) Biology of disease: role of secretory phospholipase A2 in the pathobiology of disease. Lab Invest 55:391–404
Van den Bosch H (1980) Intracellular phospholipases A. Biochim Biophys Acta 604:191–246
Verger R, Ferrato F, Mansbach CM, Pieroni G (1982) Novel intestinal phospholipase A2: purification and some molecular characteristics. Biochemistry 21:6883–6889
Vonkeman H, Van Dorp DA (1968) The action of prostaglandin synthetase on 2-arachidonyl-lecithin. Biochim Biophys Acta 164:430–432
Wassef MK, Lin YN, Horowitz MI (1978) Phospholipid-deacylating enzymes of rat stomach mucosa. Biochim Biophys Acta 528:318–330
Wilson DE, Phillips C, Levine RA (1971) Inhibition of gastric secretion in man by prostaglandin A1. Gastroenterology 61:201–206
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tatsumi, H., Tojo, H., Senda, T. et al. Immunocytochemical studies on the localization of pancreatic-type phospholipase A2 in rat stomach and pancreas, with special reference to the stomach cells. Histochemistry 94, 135–140 (1990). https://doi.org/10.1007/BF02440179
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02440179