Abstract
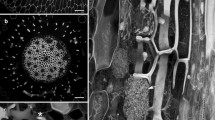
The cell wall of Cobaea scandens seed hairs developed in a characteristic sequence, with the deposition of a cellulose thread onto a pectic swelling layer was the final event. The cellulose thread was intracellularly accompanied by a band of 10–18 microtubules. During the formation of the swelling layer the microtubules were homogeneously distributed; they ran circumferentially normal to the cell axis. When cellulose-thread formation started, the microtubules became arranged in a helical band. The density of the microtubules varied during the different phases of development. The highest density was observed before cellulosethread formation and ranged from 6–15 μm·μm-2. The length of the microtubules, 20–30 μm, was determined by direct measurements, as well as estimated from the total microtubular length in a given area and the counted free ends. With the indirect immunofluorescence technique the microtubules of the band stained inhomogeneously. Those which were located at the edges of the band fluoresced more intensely than those of the central part. Attempts to visualize actin filaments in the hair cells with rhodaminyl-conjugated phalloidin resulted in a homogeneous staining of the area of the microtubular band, indicating that actin filaments may be present in this region. Though, in thin sections and dry-cleaved cells, filamentous structures were observed between the microtubules, caution is expressed that the observed fluorescence was, indeed, due to actin filaments. The role of the filamentous structures is discussed with respect to formation and maintenance of the microtubular band. Microtubules apparently did not cross coated pits which were visualized in the plasma membrane through the dry-cleaving technique.
Similar content being viewed by others
Abbreviations
- IFT:
-
indirect immunofluorescence technique
- RP:
-
rhodaminyl-conjugated phalloidin
- SEM:
-
scanning electron microscopy
References
Clayton, L., Lloyd, C.W. (1985) Actin organization during the cell cycle in meristematic plant cells. Actin is present in the cytokinetic phragmoplast. Exp. Cell Res. 156, 231–238
Dustin, P. (1984) Microtubules. Springer, Berlin Heidelber New York
Falconer, M.M., Seagull, R.W. (1985) Immunofluorescent and Calcofluor white staining of developing tracheary elements in Zinnia elegans L. suspension cultures. Protoplasma 125, 190–198
Frey-Wyssling, A. (1959) Die pflanzliche Zellwand. Springer, Berlin Heidelberg
Gunning, B.E.S., Hardham, A.R. (1982) Microtubules. Annu. Rev. Plant Physiol. 33, 651–698
Hardham, A.R., Gunning, B.E.S. (1978) Structure of cortical microtubule arrays in plant cells. J. Cell Biol. 77, 14–34
Heath, I.B., Seagull, R.W. (1982) Orlented cellulose fibrils and the cytoskeleton: a critical comparison of models. In: The cytoskeleton in plant growth and development, pp. 163–182, Lloyd, C.W., ed. Academic Press, London New York
Hepler, P.K., Fosket, D.E. (1971) The role of microtubules in vessel member differentiation in Coleus. Protoplasma 72, 213–236
Herth, W., Hausser, I. (1984) Chitin and cellulose fibrillogenesis in vitro and their experimental alteration. In: Structure, function, and biosynthesis of plant cell walls (Proc. Annu. Riverside Symp. in Botany) vol. VII, pp. 89–119, Dugger, C.W., Bartnicki-Garcia, S., eds. University of California, Riverside, Am. Soc. Plant Physiol., Rockville, Md., USA
Hoch, H.C., Staples, R.C. (1983) Visualization of actin in situ by rhodamine-conjugated phalloin in the fungus Uromyces phaseoli. Eur. J. Cell Biol. 32, 52–58
Nothnagel, E.A., Barak, L.S., Sanger, J.W., Webb, W.W. (1981) Fluorescence studies on modes of cytochalasin B and phallotoxin action on cytoplasmic streaming in Chara. J. Cell Biol. 88, 364–372
Quader, H. (1983) Morphology and movement of cellulose synthesizing (terminal) complexes in Oocystis solitaria: evidence that microfibril assembly is the motive force. Eur. J. Cell Biol. 32, 174–177
Quader, H., Herth, W., Ryser, U., Schnepf, E. (1986) Cytoskeletal elements in cotton seed hair development in vitro: on their possible regulatory role in cell wall organization. Protoplasma (in press)
Quader, H., Robinson, D.G. (1981) Oocystis solitaria: a model organism for understanding the organization of cellulose synthesis. Ber. Dtsch. Bot. Ges. 94, 75–84
Robinson, D.G., Quader, H. (1980) Structure, synthesis, and orientation of microfibrils: VII. Microtubule reassembly in vivo after cold treatment in Oocystis and its relevance to microfibril orientation. Eur. J. Cell Biol. 21, 229–230
Robinson, D.G., Quander, H. (1982) The microtubule-microfibril syndrome. In: The cytoskeleton in plant growth and development, pp. 109–126, Lloyd, C.W., ed. Academic Press, London New York
Ryser, U. (1985) Differentiating cotton fibres: a model system to study the biosynthesis of cell wall constituents and the terminal differentiation of plant cells. Eur. J. Cell Biol. 39, 236–256
Schnepf, E. (1974) Microtubules and cell wall formation. Port Acta Biol. Ser. A 14, 451–462
Schnepf, E., Deichgräber, G. (1983) Structure and formation of fibrillar mucilages in seed epidermis cells. I. Collomia grandiflora (Polemoniaceae). Protoplasma 114, 210–221
Simmonds, D., Setterfield, G., Brown, D.L. (1983) Organization of microtubules in dividing and elongating cells of Vicia hajastana Grossh. in suspension culture. Eur. J. Cell Biol. 32, 59–66
Traas, J.A. (1984) Visualization of the membrane bound cytoskeleton and coated pits of plant cells by means of dry cleaving. Protoplasma 119, 212–218
Traas, J.A., Braat, P., Derksen, J.W. (1984) Changes in microtubule arrays during the differentiation of cortical root cells of Raphanus sativus. Eur. J. Cell Biol. 34, 229–238
Wiche, G. (1985) High-molecular-weight microtubule associated proteins (MAPS): a ubiquitous family of cytoskeleta connecting links. Trends Biochem. Sci. 10, 67–70
Wick, S.M., Seagull, R.W., Osborn, M., Weber, K., Gunning B.E.S. (1981) Immunofluorescence microscopy of organized microtubule arrays in structurally stabilized meristematic plant cells. J. Cell Biol. 89, 685–690
Wulf, E., Deboben, A., Bautz, F.A., Faulstich, H., Wieland, Th. (1979) Fluorescent phallotoxin, a tool for the visualization of cellular actin. Proc. Natl. Acad. Sci. USA 76, 4498–4502
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Quader, H., Deichgräber, G. & Schnepf, E. The cytoskeleton of Cobaea seed hairs:. Planta 168, 1–10 (1986). https://doi.org/10.1007/BF00407002
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00407002