Abstract
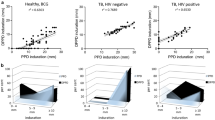
Some 840 bacille Calmette-Guérin (BCG)-vaccinated healthy controls and tuberculosis patients from two Chinese hospitals were submitted to comparative skin tests with purified protein derivative of tuberculin (PPD; as reference) and with the antigen complex A60 from Mycobacterium bovis BCG. In a first trial, including 581 persons (185 healthy juveniles, 180 healthy adults and 216 tuberculosis patients), a limited dose of A60 (1μg) was used. Performance of the A60 test was similar to that of 5 I.U. PPD for controls (cut-off values=5 mm induration diameter), but lower than that seen for tuberculosis patients (10 mm cut-off values). A second survey was conducted on 259 persons (109 recently revaccinated healthy persons, considered as tuberculin-negative in the first trial, and 150 tuberculosis patients), using a higher dose of A60 (2 μg) and the same dose of PPD (5 I.U.). Similar results were obtained with the two tests in all cases, thus supporting the possibility of PPD replacement by A60 in cutaneous testing. The pattern of induration diameter distribution in healthy subjects who took part in the first testing round (64% positively rate) was displaced to the inactivity side (with a peak at 5 to 9-mm diameter), in comparison with the second round (90% positivity rate and peak at 10–14 mm). This indicates a progressive fading of cellular immunity reactions after BCG vaccination. In tuberculosis patients, no correlation was found among the following three parameters: positivity at cutaneous testing (with PPD or A60), titer of anti-A60 mycobacterial immunoglobulins in blood (IgG titer higher than cut-off line) and presence of mycobacteria in sputum.
Similar content being viewed by others
Abbreviations
- OT :
-
Old tuberculin
- PPD :
-
purified protein derivative
- ST :
-
sensitins
- CMN :
-
Bacteria of genera Corynebacterium, Mycobacterium, Nocardia
- BCG :
-
Calmette-Guerin strain of M. bovis
- TMA :
-
thermostable macromolecular antigen complexes
- A60 :
-
the TMA complex of M. bovis and M. tuberculosis
- A36 :
-
the TMA complex of M. paratuberculosis
- A7 :
-
the TMA complex of M. leprae
- ELISA :
-
enzyme-linked immunosorbent assay
References
Amicosante M, Paone G, Ameglio F, Piccolella E, Bianchi E, Bisetti A, Luisetti M, Richeldi L, Saltini C (1993) Antibody repertoire against the A60 antigen complex during the course of pulmonary tuberculosis. Eur Respir J 6:816–822
Baelden MC, Vanderelst B, Dieng M, Prignot J, Cocito C (1990) Serological analysis of human tuberculosis by an ELISA with mycobacterial antigen 60. Scand J Infect Dis 22:63–73
Barksdale L, Kim KD (1977) Myobacterium. Bacteriol Rev 41:217–372
Beschin A, Brijs L, De Baetselier P, Cocito C (1991) Mycobacterial proliferation in macrophages is prevented by incubation with lymphocytes activated in vitro with a mycobacterial antigen complex. Eur J Immunol 21:793–797
Benoit C, Beschin A, Desmecht M, Dekeyser P, Cocito C (1989) Delayed hypersensitivity reactions by the mycobacterial antigen A60 and cutaneous testing in tuberculosis. Med Microbiol Immunol 178:105–112
Boyden SV, Sorkin E (1956) Antigens of Mycobacterium tuberculosis. Adv Tuberc Res 7:17–51
Closs O, Harboe M, Axelsen NX, Bunch-Christensen K, Magnusson M (1980) The antigens of Mycobacterium bovis strain BCG, studied by crossed immunoelectrophoresis: a reference system. Scand J Immunol 12:249–263
Cocito C (1991) Properties of the mycobacterial antigen complex A60 and its applications to diagnosis and prognosis of tuberculosis. Chest 100:1687–1693
Cocito C, Vanlinden F (1986) Preparation and properties of antigen 60 from Mycobacterium bovis BCG. Clin Exp Immunol 66:262–272
Cocito C, Baelden MG, Benoit C (1987) Immunological properties of antigen 60 and BCG. Induction of humoral and cellular immunity reactions. Scand J Immunol 25:579–585
Collins FM (1974) Vaccines and cell-mediated immunity. Bacteriol Rev 38:371–402
Collins FM, MacKaness GB (1970) The relationship of delayed hypersensitivity to acquired antituberculous immunity. I. Tuberculin sensitivity and resistance to reinfection in BCG-vaccinated mice. Cell Immun 1:253–265
Collins FM, MacKaness GB (1970) The relationship of delayed hypersensitivity to acquired antituberculous immunity. II. Effect of adjuvant on the allergenicity and immunogenicity of heatkilled tubercle bacilli. Cell Immun 1:266–275
Council of Europe. European Pharmacopaeia Commission (1985) Tuberculini bovine derivatum proteinosum purificatum. Document PA/PH/Exp3/T(84)54, Strasbourg
Crowle AJ (1958) Immunizing constituents of the tubercle bacillus. Bacteriol Rev 22:183–213
Cummins CS (1962) Chemical composition and antigenic structure of cell walls of Corynebacterium, Mycobacterium, Nocardia, Actinomyces and Arthrobacter. J Gen Microbiol 28:35–50
Daniel TM (1980) The immunology of tuberculosis. Clin Chest Med 1:189–201
Daniel TM, Janicki BW (1978) Mycobacterial antigens: a review of their isolation, chemistry and immunological properties. Microbiol Rev 42:84–113
De Kesel M, Gilot P, Coene M, Cocito C (1992) Composition and immunological properties of the protein fraction of A36, a major antigen complex of Mycobacterium paratuberculosis. Scand J Immunol 36:811–821
Dobbelaer R, O'Reilly LM, Génicot A, Haagsma J (1983) The potency of bovine PPD tuberculins in guinea-pigs and in tuberculous cattle. J Biol Stand 11:213–221
Edwards PQ, Edwards LB (1960) Story of the tuberculin test from an epidemiologic viewpoint. Am Rev Respir Dis 81 [Suppl]:1–47
Fabre I, L'Homme O, Bruneteau M, Michel G, Cocito C (1986) Chemical composition of antigen 60 from Mycobacterium bovis BCG. Scand J Immunol 24:591–602
Gilot P, De Kesel M, Coene M, Cocito C (1992) Induction of cellular immune reactions by A36, an antigen complex of Mycobacterium paratuberculosis. Comparison of A36 and Johnin components. Scand J Immunol 36:811–821
Goren MB (1982) Immunoreactive substances of mycobacteria. Am Rev Respir Dis 125 [Suppl]:50–69
Haagsma J, O'Reilly LM, Dobbelaer R, Murphy TM (1982) A comparison of the relative potencies of various bovine PPD tuberculins in naturally infected tuberculous cattle. J Biol Stand 10:273–284
Harboe M (1981) Antigens of PPD, old tuberculin and autoclaved M. bovis BCG studied by crossed immunoelectrophoresis. Am Rev Respir Dis 124:80–87
Harboe M, Closs O, Bjorvatn B, Bjune G (1977) Antibodies against BCG antigen 60 in mycobacterial infection. Br Med J 2:430–433
Hubbard RD, Flory CM, Cocito C, Collins FM (1992) Immunization of mice with antigen A60 from M. bovis BCG. Clin Exp Immun 88:129–131
Journal officiel des Communautés européennes (1978) Publication no. L15, 34–41
Kanai K, Youmans GP, Youmans AS (1960) Allergenicity of intracellular particles, cell walls, and cytoplasmic fluid from Mycobacterium tuberculosis. J Bacteriol 80:615–621
Kardjito T, Handoyo I, Grange JM (1982) Diagnosis of active tuberculosis by immunological methods. 1. The effect of tuberculin reactivity and previous BCG vaccination on the antibody levels determined by ELISA. Tubercle 63:269–274
Kim TC, Blackman RS, Heatwole KM, Kim T, Rochester DF (1984) Acid-fast bacilli in sputum smears of patients with pulmonary tuberculosis. Prevalence and significance of negative smears pretreatment and positive smears post-treatment. Am Rev Respir Dis 129:264–268
Kuwabara S (1975) Purification and properties of tuberculin-active protein from Mycobacterium tuberculosis. J Biol Chem 251:2556–2562
Landi S, Held HR, Gupta KG (1975) The multi-facets of tuberculin standardization. Dev Biol Stand 29:293–411
Lefford MJ (1975) Delayed hypersensitivity and immunity in tuberculosis. Am Rev Respir Dis 111:243–246
Lefford MJ (1975) Transfer of adoptive immunity to tuberculosis in mice. Infect Immun 11:1174–1181
Lesslie IW (1976) Correlation of biological potency of human and bovine tuberculin PPDs in guinea-pigs, cattle and man. J Biol Stand 4:39–42
Lipsky BA, Gates J, Tenover FC, Plorde JJ (1984) Factors affecting the clinical value of microscopy for acid-fast bacilli. Rev Infect Dis 6:214–222
Lurie MB (1942) Studies on the mechanism of immunity in tuberculosis: the fate of tubercle bacilli ingested by mononuclear phagocytes derived from normal and immunised animals. J Exp Med 75:247–267
Mackaness GB (1962) Cellular resistance to infection. J Exp Med 116:381–406
Mackaness GB (1964) The immunological basis of acquired cellular resistance. J Exp Med 120:105–120
Mackaness GB (1968) The immunology of antituberculous immunity. Am Rev Respir Dis 97:337–344
Mackaness GB, Blanden RV (1967) Cellular immunity. Prog Allergy 11:89–140
Neiburger RG, Youmans GP, Youmans AS (1973) Relationship between tuberculin hypersensitivity and cellular immunity to infection in mice vaccinated with viable attenuated mycobacterial cells or with mycobacterial ribonucleic acid preparations. Infect Immun 8:42–47
Organisation Mondiale de la Santé (Genève) (1979) Listes des étalons biologiques internationaux, des préparations biologiques internationales de référence et des réactifs biologiques internationaux de référence
Palmer CE, Edwards LB, Hop wood L, Edwards PQ (1959) Experimental and epidemiologic basis for the interpretation of tuberculin sensitivity. J Pediatr 5:413–429
Pharmacopée européenne (1982) Deuxième édition. Tuberculini derivatum proteinosum purificatum ad usum humanum. Maisoneuve S.A., Sainte Ruffine, Monog. 151
Seibert FB (1949) The isolation of three different proteins and two polysaccharides from tuberculin by alcohol fractionation. Their chemical and biological properties. AM Rev Tuberc 59:86–101
Seibert FB, Glenn JT (1941) Tuberculin purified protein derivative. Preparation and analysis of a large quantity for standardization. Am Rev Tuberc 44:9–25
Sindic CJM, Boucquey D, Van Antwerpen MP, Baelden MC, Laterre C, Cocito C (1990) Intrathecal synthesis of antimycobacterial antibodies in patients with tuberculous meningitis. An immunoblotting study. J Neurol Neurosurg Psychiatry 53:662–666
WHO Expert Committee on Biological Standardization (1968) Requirement for tuberculins, WHO Technical Report Series 384. WHO, Geneva, pp 23–41
Wijsmuller G, Bardine A, Rust P, Seligman EB, Armstrong FC (1975) Evaluation of the animal assay of tuberculin intended for use in humans. I. Reproductibility. J Biol Stand 3:51–63
Youmans GP (1975) Relation between delayed hypersensitivity and immunity in tuberculosis. Am Rev Respir Dis 111:109–118
Youmans GP (1979) Development of delayed (tuberculin) hypersensitivity in tuberculosis. In: Youmans GP (ed) Tuberculosis. Saunders, Philadelphia, pp 209–224
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zou, Y.L., Zhang, J.D., Chen, M.H. et al. Comparative cutaneous testing with purified protein derivative and the antigen complex A60 in vaccinated subjects and tuberculosis patients. Med Microbiol Immunol 184, 9–15 (1995). https://doi.org/10.1007/BF00216784
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00216784